Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 10ALQ
Related questions
Question
100%
| Runs | [S2O8 2-], M | [I–], M | [S2O3 2-], M | Reaction time,s | Rate, M/s |
|---|---|---|---|---|---|
| 1 | 49.10 | ||||
| 2 | 106.10 | ||||
| 3 | 197.44 | ||||
| 4 | 69.20 | ||||
| 5 | 53.05 |
please complete the table
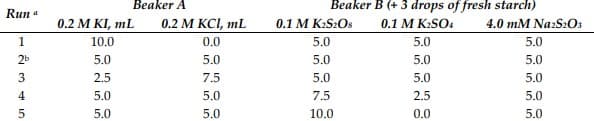
Transcribed Image Text:Beaker A
Beaker B (+ 3 drops of fresh starch)
Run a
0.2 M KI, mL
0.2 М KСL, mL
0.1 M K:S2OS
0.1 M K:SO4
4.0 mM Na:S2O3
1
10.0
0.0
5.0
5.0
5.0
2b
5.0
5.0
5.0
5.0
5.0
2.5
7.5
5.0
5.0
5.0
4
5.0
5.0
7.5
2.5
5.0
5.0
5.0
10.0
0.0
5.0
เก่ เก่ เก่
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning