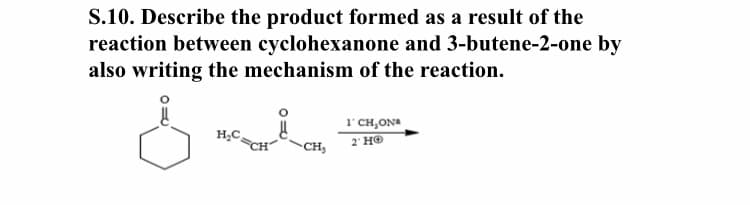
S.10. Describe the product formed as a result of the reaction between cyclohexanone and 3-butene-2-one by also writing the mechanism of the reaction. 1'CH,ON H;C, 2 HO CH,
Q: Give the major organic product(s) of the reactions of 1-methyl-2-deuteriocyclohexene (D =?H) with…
A:
Q: In attempting to make 2–bromo–2–methyl–propane from 2–methyl-propene, as shown below, you used a…
A: Interpretation - We have to tell about the mechnaism of unwanted by-product formation in the avobe…
Q: V. Give the structures of the products expected from the reaction of 3-methyl-1-pentyne with each of…
A:
Q: The reaction of methylpropene with HBr, under radical conditions, gives twO intermediates. Propose a…
A: Given:-
Q: 2-bromobutane can be prepared in the lab by using organic chemicals and bromine in inert solvent. a)…
A: The solution is as follows:
Q: Propane reacts with Br2 to form 2-bromobutane in the presence of uv light. Produce the minor…
A: Alkanes when react with halogen in the presence of UV light, haloalkane is formed. The formation of…
Q: Provide a series of synthetic steps by which (CH3)2C CH2 could be prepared from 2-methylpropane.
A: Interpretation- To provide the synthesis steps by which (CH3)2C=CH2 could be prepared from…
Q: A ketone undergoes acid-catalyzed bromination, acid-catalyzed chlorination, racemization, (see page…
A:
Q: Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane…
A:
Q: Reaction of this bicycloalkene with bromine in carbon tetrachloride gives a trans dibro- mide. In…
A:
Q: Give the major organic product(s) of the reactions of 1-methyl-2-deuteriocyclohexene (D = 'H) with…
A: (a) The Anti-Markovnikov synthesis addition of H2 at C=C occurs.
Q: O3 + НО—ССH-CH + CH;CCH,CH(CHO), CO2 но, ?? Zn
A:
Q: What will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: O3 CO2 + HO-CCH,CH CH;CCH,CH(CHO)2 ?? Zn
A:
Q: Please explain the mechanism for the Replacement of OH by halogen in reaction with HCl or HBr (SN1…
A:
Q: Write the etherification reactions of isopropyl bromide and 2-bromo-1-nitropropane with phenol .…
A:
Q: Part A: Please explain in detail the mechanism of catalytic hydrogenation when applied to the…
A:
Q: Problems 1 and 3 for part a (3-methyl-3-pentanol with phosphoric acid)
A: The alcohol taken in part A) is 3-Methyl-3-pentanol. The alkenes formed during the experiment are as…
Q: When 2-bromobutane is reacted with CH3O-, two alkene products, namely 2-butene and 1-butene are…
A: The reaction of 2-Bromo butane with MeO^- gives 1-butene and 2-butene. This reaction is passing…
Q: M5 Explain the different stages of reaction, reaction mechanisms, conditions of reactions, (for the…
A: M5)A reaction takes place in different stages for example lets take a second order reaction it…
Q: 2. a. Write the complete reaction equation along with all the products that might be formed if…
A: Elimination reaction are reaction where a functional group is lost to form alkene
Q: Suppose you were told that each reaction is a substitution reaction, but you were not told the…
A: SN1 reactions are most speedy with tertiary alkyl halides, they happen more gradually with secondary…
Q: What would the major organic reaction product be from the reaction of 1-bromo-1-methylcyclopentane…
A:
Q: a. Write two reaction paths for the production of monochlorodecane (C10H21CI) by reaction of Decane…
A: In the presence of sunlight, a radical substitution reaction takes place and monochlorodecane is…
Q: Keeping in mind that carbocations have a trigonal planar structure, (a) write a structure for the…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: In the reaction scheme below, the furan A is converted to the thiophene B via a diketone. Write a…
A: Formation of a Diketone:
Q: Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Draw…
A: The reactants given are iodoethane and below alkoxide
Q: OH H,SO4 H3C-CH-CH2-CH3 H3C-CH=CH-CH3 H20 Acid-catalyzed dehydration of 2-butanol yields 2-butene as…
A: Given reaction is : Given incomplete mechanism is : Complete the curved arrow mechanism = ?
Q: y isomers can be formed in each case? (2) Addition of HBr to 3,3-dimethyl-1-butene gives a mixture…
A: The addition of particular group without any substitution. For eg., HBr addition in a double bond.
Q: Adding 1 mole of hydrogen chloride (HCI) to 1,3-octadiene gives two products. Give their structures,…
A: 1,3-octadiene reacts with 1 mol HCl to produce two products, say A and B. Here compounds A and B…
Q: The reaction of (2S)-2-chloro-3-methylpentane with sodium iodide yields two products:…
A:
Q: Provide the E2 mechanism for β-elimination reaction employing 2-chloro-2-methylbutane to prepare…
A: For E2 reaction, If base is hindred so, Hoffman alkene is formed , less stable alkene acts as a…
Q: 3.3 8 Give the reaction mechanism and name the products for the reaction of 2-bromobutane with a hot…
A: Given- Reactant- 2-bromobutane ,hot ethanolic solution of KOH ->Here OH is nucleophile.
Q: 2. Provide the final product for each of the following reactions: a) 1-methylcyclohexene reacts with…
A: For the all the given reactions we can expect alcohol as the product and the position of OH group is…
Q: 3 Give mechanisms for the acid-catalyzed and base-promoted alpha-halogenation ofketones. Explain why…
A: The mechanisms for the acid-catalyzed alpha-halogenation of ketones is,
Q: (d) Ozonolysis of 3,4-dimethyl-3-hexene gives compound D. (1) Write the reaction step for the…
A:
Q: Dihydroxylation of an alkene can be carried out with H2O2 in HCO2H. In this reaction,…
A:
Q: Show the process of synthesizing 2-methyl2-cyclohexenone from 2-methyl cyclohexanone. Write down the…
A: The solution is as follows:
Q: 2а. with ammonia. Provide a detailed, stepwise mechanism for the reaction of propanoyl chloride 2b.…
A: Answer:- This question is answered by using the simple concept of chemical reaction of organic…
Q: 3. Write in the product of 3-bromo-4-chloro-1-pentyne with the following reagents: 1. BH, 2. HO2,…
A:
Q: Suppose you are told that each reaction is a substitution reaction but are not told the mechanism.…
A:
Q: In the reaction below, 3-Buten-2-one reacts with 2,3-Dimethyl-1,3-butadieneand 1,3-Butadieneat 140°C…
A: A question based on Diel's-Alder reaction, which is to be accomplished.
Q: 4) Provide the structure of the major organic product of the reactions below. (CH;CH,),N NaOCH CH,…
A: In this question, we want to draw the structure of major product. Reagent and starting material are…
Q: write the reaction of the action of methyl-2, bromo-2-propane with hot KOH solution. a) Name the…
A:
Q: Treating a hindered alkene such as 2-methyl-2-butene with BH3:THF leads to the for- mation of a…
A: Explanation- Disiamylborane (bis(1,2-dimethylpropyl)(borane, Sia2BH) or (bis (3-methyl-2-butyl)…
Q: Write the synthesis of the target compounds by starting from each benzene given in the following…
A: (“Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Some 2-methyl-2-butene may be produced in the reaction as a by-product. Give a mechanism for its…
A: The formation of tert-pentyl chloride is formed by the reaction of tert-pentyl alcohol with HCI. The…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- What organic product would you obtain from reaction of 1-pentanol with CrO3, H2O, H2SO4?A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsDisiamylborane adds only once to alkynes by virtue of its two bulky secondary isoamylgroups. Disiamylborane is prepared by the reaction of BH3 # THF with an alkene.(a) Draw the structural formulas of the reagents and the products in the preparation ofdisiamylborane.(b) Explain why the reaction in part (a) goes only as far as the dialkylborane. Why isSia3B not formed?
- Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsProvide the E2 mechanism for β-elimination reactionemploying 2-chloro-2-methylbutane to prepare 2-methyl-2-butene and 2-methyl-1-butene reaction. Use the actual structures of thereactants and products. Explain which of the alkenes is the major product ofthis reaction ?Predict the organic products of the reaction of 2 butene with each reagent. Be sure to indicate stereochemistry and regioselectivity where appropriate. a. Br2 in H2O b. Hg(OAc)2, H2O c. Product from (b) + NaBH4
- When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.Compounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.What is the reaction when trans-2-butene reacts with 1.KMnO4/-OH in cold 2. hotKMnO4/-OH 3.HBr in the presence of peroxide
- 2-methyl-1-propanol heated with acidic K2Cr2O7 reactionWrite equations for the reaction of 1-butanol with reagent. Where you predict no reaction, write NR. Q) K2Cr2O7, H2SO4, H2O, heatWhat will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carriedout ina. dichloromethane? b. water? c. ethyl alcohol? d. methyl alcohol?

