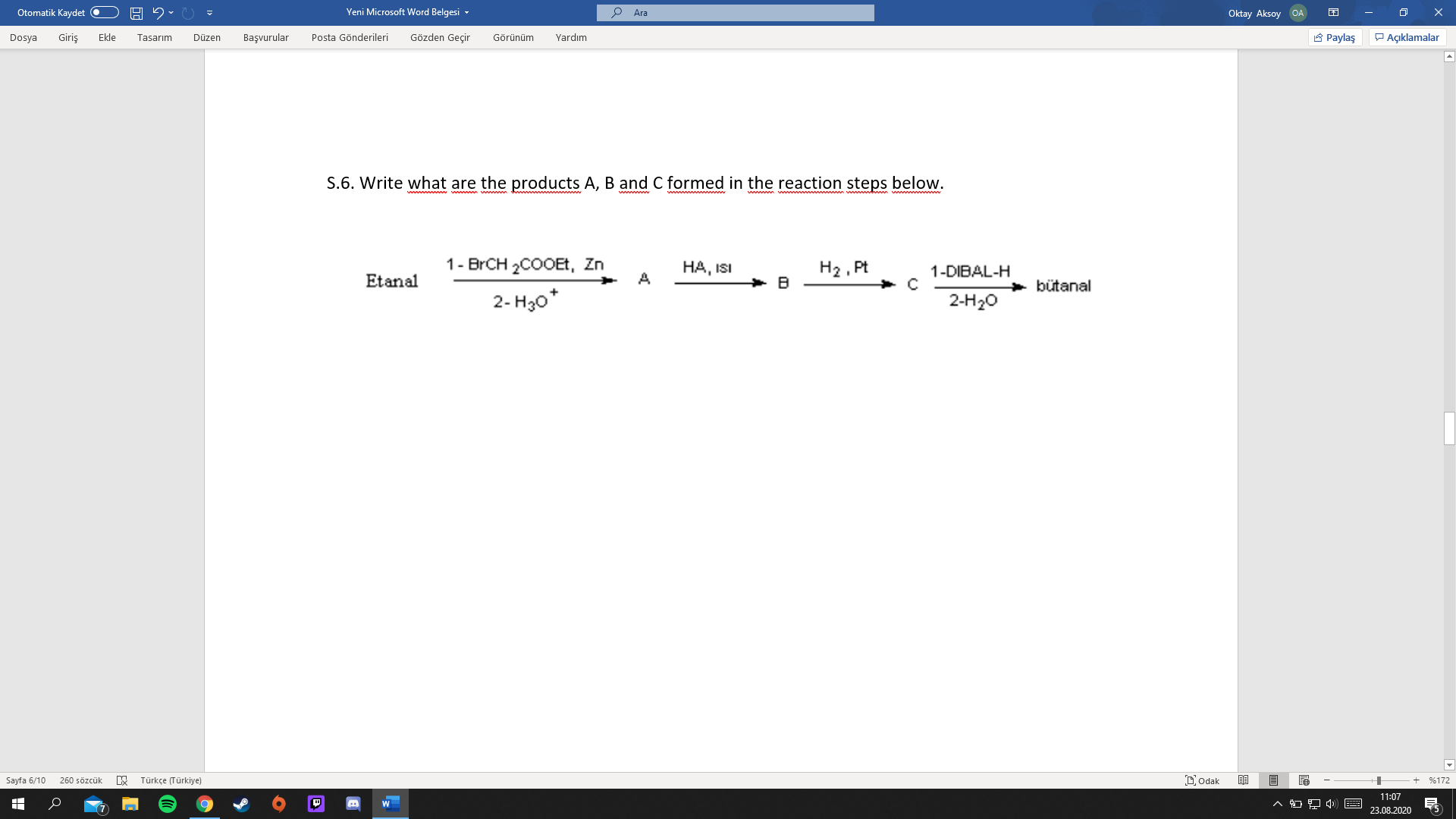
S.6. Write what are the products A, B and C formed in the reaction steps below. www 1- BRCH 2COOEt, Zn H2 , Pt B НА, ISI 1-DIBAL-H Etanal A bütanal 2- H30* 2-H20
Q: sed on the reaction scheme below, draw the structure for compounds D, E, F (i) LIAIH4 in ether (ii)…
A:
Q: 5.) What does the reaction below describe? O2(g) + hv --------> O2+ + e- Group…
A: The reaction can be described by
Q: (c) Write the product(s) for the following reaction with supportive mechanism : ŞiMez KH →? Bu, Нех…
A: Here the given reaction is an example of Peterson Olefination. So, in this reaction alkene is…
Q: . (a) Azulene, shown below, has the ability to react with both electrophiles as well as…
A: The solution of the question is given below:
Q: Which compound shows the highest rate of reaction when shaken vigorously with ethanolic silver…
A:
Q: (d) Propose a reaction mechanism to account for the following reaction. AICI3
A: In this question we have to tell the mechanism of the given reaction.
Q: The order of decreasing rectivity towards electrophilic reagent for the following: (a) Benzene (b)…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Criteria for satisfactory score Reactants, products, and reagents that complete a reaction scheme…
A: A. At first the alcohol gets converted into chloride on reaction with SOCl2. Then it forms Grignard…
Q: Give the major product for the reaction below. A H20 HCI cold, dilute B KMNO4 KMNO4, OH, heat E HBr…
A: The reactions are summarized as: The acidic hydration of alkenes takes place according to…
Q: Give the major product for the reaction below. A HBr H2 F Ni KMNO4, OH, heat H* KMNO4 Br2 E cold,…
A: Given,
Q: Give the major product for the reaction below. A H20 HCI cold, dilute B KMNO4 KMNO4, OH, heat E HBr…
A: Answer of the question given below,
Q: H3C. CH3 ? H3C. CH3 но снз 27. The reaction shown above may be best carried out by reacting…
A:
Q: 2. A). Give the structure for the compounds A through E. (There is a mistake in the structure of the…
A:
Q: write mechanism step by step for the reaction ?
A: The given reaction is
Q: Predict the product formed when the compound shown below undergoes a reaction in the presence of…
A: The product of the above transformation is given below
Q: 23. Congo Red is a synthetic dye whose use was later abandoned because of its carcinogenic…
A: (23) answer: Correct option (b) From Retrosynthesis analysis we can say, Nelson can synthesize…
Q: 17 Give the missing reactant(s), product(s) or reagent(s) ONLY (no mechanisms). (Non-anonymous…
A: T his reaction have two parts . First is the reduction of H2/pd c (1 eq) The Lindlar catalyst…
Q: 6) Consider the following reactions scheme below. NNH- H. HgCB Step II Step I HO Step II H HCN…
A:
Q: Give the major product for the reaction below. A H20 HCI F cold, dilute B KMNO4 KMNO4, OH, heat E…
A:
Q: Provide a plausible arrow pushing mechanism for the reaction below. OH LOME TSOH, H20 OMe
A:
Q: NEN Semester One Examinations, 2020 Question A6 Show how you would complete the following…
A: Soln-
Q: In the reaction scheme below, the furan A is converted to the thiophene B via a diketone. Write a…
A: Formation of a Diketone:
Q: Predict the products of reaction of pent-1-yne with the following reagents.(a) 1 equivalent of HCl…
A: “Since you have asked multiple questions, we will solve the first three question for you. If you…
Q: Which step in the following reaction will cause the proposed synthesis to fail? A B .CI NBS -Br…
A:
Q: Question is attached
A: These are the organic reaction, in which from two or more reactants it gives now product by breaking…
Q: 1. Show the major products of reactions with 1-pentyne, with stereochemistry where applicable, for…
A:
Q: 1. Show the major products of reactions with 1-pentyne, with stereochemistry where applicable, for…
A: Since you have asked a question with multiple subparts, we will solve the first three subparts for…
Q: (c) Predict the product(s) for the following reaction with supporting mechanism. i) SeO2 Ag diovone
A: Selenium dioxide, SeO2 is an oxidizing agent generally employed in the allylic oxidation of alkenes…
Q: Give the major product for the reaction below. A HBr H2 F Ni KMNO4, OH, heat B H* KMNO4 Br2 E cold,…
A: Given that : We have to give all the products formed for the reactions :
Q: 6.Give the major organic product(s) of the following reactions. nily CH, Н.С Н.С a. + H2O 1.…
A: The major product formed in a is shown below.
Q: The following reactions have been used to synthesize dieldrin and aldrin (named for Diels and…
A: This suggest that this reaction would be diels alder reaction and the molecule X is an alkene,…
Q: 5. Predict the product of the following reactions. Provide the major organic product from cach. H=…
A: Yes, all answers are correct. a) this is the O-alkylation reaction, in which strong base NaH…
Q: The structures of alkenes can affect their reactivity in the reaction with H-Br. H;C H H;C H H H `H…
A: Given : We have tell the ease of alkene to accept a proton from HBr.
Q: Predict the product of the reaction shown here. 1. BH3 THF (excess) ? 2. NaOH, H2O2, Н,О…
A: Addition of BH3 on double bond of alkene takes place according to anti-markovnikov rule. In this…
Q: Which structure shows correctly the product of the reaction below? н. В -BUOK b. d.
A: In the elimination reaction of organic compounds, two substituents are removed from the organic…
Q: (a) Give starting materials (including molar ratios) for synthesis of compound Z. Ph EIO2C OEt F3C…
A: This is an example of Hantz pyridine synthesis.
Q: 12:53 ul 4G O 2. Show a reaction scheme with all the reactants and reagents of the nitration…
A: Dear student this question is related to nitration of bromo benzene.
Q: Provide the best combination of reagents for the following transformation. NMe NME2 O 1. H+,…
A:
Q: Provide the products for the below reactions: HgSO4 H,SO4 ВНЗ, ТHF H2O2, OH", H2O
A: HgSO4 and aqueous dilute H2SO4 is used for alkyne hydration. Alkynes hydration is much harder than…
Q: Provide a plausible arrow pushing mechanism for the reaction below. You may abbreviate the…
A: The reactions follow (2π+4π ) cyclo addition Diels elder reaction. The first step shows Diels -alder…
Q: II. Predict the products of the ff: elimination reactions and show the mechanisms involv HO H2SO4…
A:
Q: Give the molecular structures of reagent or products for the following chemical reactions: (i)…
A: We have to predict the molecular structures of reagent or products for the the following given…
Q: 1. (pr the schemes below. B1₂ light S. Br h). Provide the following structures and reaction…
A: From the methyl cyclopentane if you add Br2 in prence of light it form alkyl bromide. After…
Q: i. TBHP, Zn(SO,CF3)2 H3C- FOCH3
A: The reactant is an compound with ether functional group, that is an alkyl and aromatic group is…
Q: Propose a reasonable synthetic route for the following reaction: II CH,CH,-C-CI Zn(Hg) (1)? (2)?…
A:
Q: 5. Consider the following reactions scheme below. NOH Step II OH Step 1 HCN A C&H5MGCI / H3O* a)…
A:
Q: i. LIAIH4 i. deprotection .CO2ME J CH2=CHCOCH3 protection ii. H30* ii. dehydration K H The synthesis…
A: Here we have to write the major product formed and reagents involved in protection , deprotection…
Hello There. I need some help. S-6
Step by step
Solved in 3 steps with 2 images

- (a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acidFor butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).10.14 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IV
- 1)2PbS + 3O2 on heating gives 2PbO + 2SO2 2)PbS + 2O2 on heating gives PbSO4 Why is it that 2 reaction shows different products? Please explain in detailI am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!please quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.

