(S208 2-), M In rate vs In [S208 2-] at constant [1-] In [S208 2-) Runs Reaction time, s Rate, M/s In rate Slope Intercept r2 value Rate order Rounded off Rate orders are typically whole numbers or fractions Runs Rate constant 1 2a 3 4 Average
(S208 2-), M In rate vs In [S208 2-] at constant [1-] In [S208 2-) Runs Reaction time, s Rate, M/s In rate Slope Intercept r2 value Rate order Rounded off Rate orders are typically whole numbers or fractions Runs Rate constant 1 2a 3 4 Average
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter32: Radiochemical Methods
Section: Chapter Questions
Problem 32.12QAP
Related questions
Question
pls complete the table below
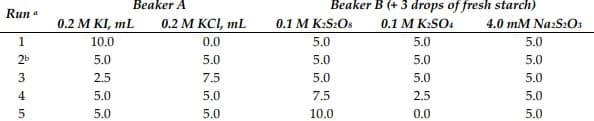
Transcribed Image Text:Beaker A
Beaker B (+ 3 drops of fresh starch)
Run a
0.2 M KI, mL
0.2 М KСL, mL
0.1 M K:S2OS
0.1 M K:SO4
4.0 mM Na:S2O3
1
10.0
0.0
5.0
5.0
5.0
2b
5.0
5.0
5.0
5.0
5.0
2.5
7.5
5.0
5.0
5.0
4
5.0
5.0
7.5
2.5
5.0
5.0
5.0
10.0
0.0
5.0
เก่ เก่ เก่
![[S208 2-], M
In rate vs In [S208 2-] at constant [I-]
In [S208 2-]
Runs
Reaction time, s Rate, M/s
In rate
Slope
Intercept
r2 value
Rate order
Rounded off
Rate orders are typically whole numbers or fractions
Runs
Rate constant
1
2a
3
4
Average](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F55815ef3-1f3f-422e-b33b-6261d3029e90%2Fe3e153dc-b5a4-433b-8204-7bc704dc0318%2Feban4zg_processed.jpeg&w=3840&q=75)
Transcribed Image Text:[S208 2-], M
In rate vs In [S208 2-] at constant [I-]
In [S208 2-]
Runs
Reaction time, s Rate, M/s
In rate
Slope
Intercept
r2 value
Rate order
Rounded off
Rate orders are typically whole numbers or fractions
Runs
Rate constant
1
2a
3
4
Average
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
