Q: CH3 H3C. „NH2 NABH(OAc)3 H3CCO2H NEt
A: We have to fill the blank with the products of the given reaction.
Q: Which rate law fits with this mechanism? 2NO --> N2O2…
A: Rate law says Rate is directly proportional to the concentration of reactants with raising the…
Q: The initial rates for the following two elimination reactions were measured under different 9.…
A:
Q: Rate = k[acetone]1[I2]0[H+]1 rate = 0.001M/ 332.26s = 3.01 x 10-6 my question is what is the…
A: Given the rate law expression of the reaction is as follows: Rate =…
Q: Answer the following questions regarding the reaction between pentene and HCI below. CI HCI а. Given…
A:
Q: The initial rates for the following two elimination reactions were measured under different…
A:
Q: 12. Show the initiation, propagation, and termination steps (6 reactions total) for the following…
A:
Q: Br NACN (2.5 ermie. n) Br NaNH2 (2eriv ) heat Br Br
A:
Q: plete the following reactions ( . H2SO3 + Ba(OH)2 > . CAH9OH + 02 > Br2 + YF3 >
A: In the balanced chemical reaction number of atoms of each element in product side is equal to number…
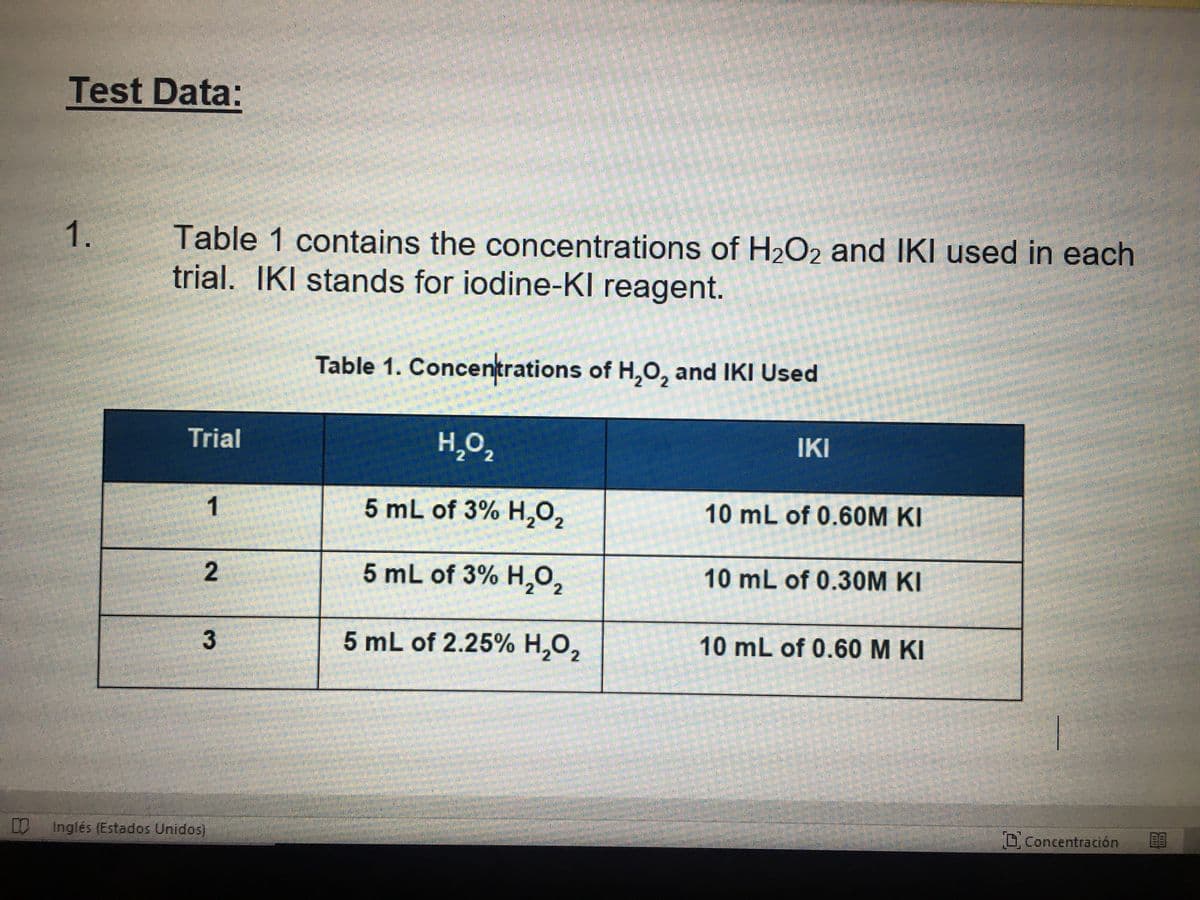
Q: Table 2: Molarity of H2O2 and KI and Reaction Rate Trial H2O2 Concentration, M KI Concentration,…
A: Trial 1 slope = 14.47 sec/ml 1/slope = 0.069 ml/sec Trial 2 slope = 24.16 sec/ml 1/slope = 0.041…
Q: rate constant
A:
Q: Add these 2 equations and obtain an overall reaction (cancel out compounds that appear in the…
A: The given two equations are as follows: (CH3)2SiCl2 +2H2O → (CH3)2Si(OH)2 +2HCl n(CH3)2Si(OH)2 →…
Q: entify all 4 intermediates from the fa A-1) drawn below (it will help if you reaction), NOTE: there…
A: Mechanism
Q: Q- The value of rate constant for reaction between terphthalic acid (0.5x10 mol/L) with HCI (0.1N )…
A: Composite reactions are the reactions that occur in more than one step or involve more than one…
Q: Which of the following step in the reaction scheme is either out of order or correct and will not…
A: Alkyl or aryl halides are prepared usually by direct halogenation. Substituted halides on benzene…
Q: (iv) What is the proportion of starting material that remains after three (3) half-lives?
A: At half life time the concentration of starting material will become half.
Q: rate law expression is R=K[i-J][H2O2[H+] because all my integers were 0, 0, 0. in my instructions…
A: Rate law is written in terms of concentration and order of reactants.
Q: I) II) R3C------H-CI R3C------H-…CI Reaction Profile Diagram Reaction Profile Diagram III) IV)…
A: In radical chlorination, the key propagation step is hydrogen abstraction.
Q: What conditions does the Wurtz-Fittig reaction requires for making biphenyl (Temp, duration, N2 or…
A: Formation of alkylated, aromatic hydrocarbons by coupling of alkyl and an aryl halides mediated by…
Q: ction rate constant be negative? 0 15 20 5 -1 5 -2 5 3 5 10 25 y = -0.042x-3.0229 R² = 0.9995 30
A: Given data set: Plot of concentration vs time. Equation of line: y= - 0.042x - 3.0229 We know that:…
Q: Reaction progress Reaction Which of the following is correct with respect to these diagrams? O A…
A: Applying concept of graph formation of E1and E2 elimination reaction.
Q: CH3 CH;CHCH,CH,CH3 „CHCH,CH,CH3 а. Cl AICI3
A:
Q: A strong base is favourable for E2 pathway. E2 reactions always involve a strong base. They need…
A: E2 elimination follows a concerted mechanism reaction in which the removal of leaving group and…
Q: An irreversible unimolecular reaction (A -> B) has a rate constant of 3.0 per second. What is the…
A: Given that value of rate constantK of an irreversible unimolecular reaction A→B is 3.0 per second.…
Q: The rate law of the elementary reversible reaction CO + 2H2 CH3OH is k [ Cco CH2 - K CcH3OH ) is…
A: Given: The reversible reaction is:CO +2H2 ⇌ CH3OH
Q: Limited Reactions Questions Please show work with parts A, B and C Thank you!
A:
Q: Cinnamoyl chloride (C9H7CIOO) can be prepared by the two-step sequence shown below. Reaction 1:…
A:
Q: Effect of Temperature Trial # Temperature Time, (sec) Rate of Reaction 1 0 °C 250. sec Click…
A: The rate of the reaction can be solved as: 1. Given information: Trial Temperature (°C)…
Q: Match
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Target Reaction: Step Reactions:
A:
Q: The rate for trial #3 is 1.5x10-3 4. The following chart includes data from a series of experiments…
A: Rate law expression gives the rate of reaction in terms of concentration of reactants each raised to…
Q: Average Reaction Rate of Reaction Time (s) Molarity Molarity of Acetone (M) of HCI (M) Molarity of…
A: Given: Order for Acetone = 1 Order for H+ (HCl) = 1 Order for Iodine = 0 Rate of reaction for…
Q: Table 2: Molarity of H2O2 and KI and Reaction Rate Trial H2O2 Concentration, M KI Concentration,…
A: trial 1 slope = 14.47 sec/ml 1/slope = 0.069 ml/sec trial 2 slope = 24.16 sec/ml 1/slope = 0.041…
Q: In rate vs In [S208 2-] at constant [I-] In [S208 2-1 Reaction time, s Rate, M/s 0.00000 0.00000…
A: Table to be completed
Q: +21 + 2HO HOI + OH Slow Fast 1-1- H0 Fast rate law that is consistent with this meche e Choice…
A: As we know, in the reaction mechanism, slow step is rate determining step (RDS). Rate is depend upon…
Q: For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be…
A: Methanol (CH3OH) is a polar protic solvent while dimethyl sulfoxide is an aprotic solvent. The polar…
Q: I analyze the reaction of Compound C with excess NAOH at 298 K. and get the following results for…
A: We plotted ln([absorbance]) on our y axis and time, t, on our x axis. The result is a straight line…
Q: 10. Heterolytic and beterogenis reactions have symmetrical reaction mechanism.
A: Welcome to bartleby !
Q: [S208 2-], M In rate vs In [S208 2-] at constant [I-] In [S208 2-] Reaction time, s Rate, M/s…
A: The given experimental data is for iodine clock reaction. The balanced equation for iodine clock…
Q: lass, we learned about radical halogenation. Instead of using 12 or C12 to halogenate propane, ou…
A:
Q: Which rate law fits with this mechanism? 2NO --> N2O2…
A: According to rate law;Rate of reaction=K{concentration of reactant}. In the given question there is…
Q: 18. Derive the rate law for the following multi-step reaction: CH4 + OH = CH3 + H2O (fast) CH3 + O2…
A: Rate law gives the relation between the rate of the reaction and the concentration of the reactants.…
Q: Calculate the reaction rate for each trial using an average of the time (in sec) for each trial
A: Average time for all Trials Trial-1 (1) 275.65 s (2) 281.31 s (3) 285.31 s Average of three values…
Q: List 2 chemical reactions that can produce chemical products ( by-products or main products) with…
A: A question about waste management, which is to be accomplished.
Q: Classify each of the following reactions: A. C2H4 (g) + 302 (g)→2CO2(g)+ 2H2O(g) B. 2H9O(s)→2H|(1) +…
A:
Q: Would the exponents, m and n, in your rate expression be larger than, smaller than, or unchanged…
A: a) A leak occured in the mouth of flask value will be unchanged as no gaseous reaction taking…
Q: || CH3CH₂CH₂CH₂COE 1. Br₂. CH3CO₂H 2. Pyridine, heat CH3CH₂CH=CHCOEt The reaction will not happen…
A:
Q: Complete these reactios, and reaction schemes by filling in the blanks reaction products. All except…
A: (A) When primary alcohol reacts with Pyrimidine chloro chromate (PCC) , it will give Aldehyde and…
Q: reactant 1 reactant 2 product 1 product 2 + + chemical reaction: N2:CO3(aq) + appearance of…
A: CuCl2 + Na2CO3 --------------> CuCO3 + 2NaCl
Table 2: Molarity of H2O2 and KI and Reaction Rate
|
Trial |
H2O2 Concentration, M |
KI Concentration, M |
Reaction Rate |
|
1 |
0.29 M |
0.40 M |
14.08 |
|
2 |
0.29 M |
0.20M |
25 |
|
3 |
0.023 M |
0.40 M |
20 |
help me with this part please, and show me all steps
- Order with respect to H2O2:
- Order with respect to KI:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- I need help with filling out the accompanying chart. Here is the data provided: Part A: The standardized HCl solution being used is 0.198 M. Trial 1: Using a volumetric pipette, you pipet exactly 20.00mL of the standardized HCl solution into a 250-mL Erlenmeyer flask. You then add 2-3 drops of phenolphthalein. Trial 2: Using a volumetric pipette, you pipet exactly 10.00mL of the standardized HCl solution into a 250-mL Erlenmeyer flask. You then add 2-3 drops of phenolphthalein. Trial 3: Using a volumetric pipette, you pipet exactly 10.00mL of the standardized HCl solution into a 250-mL Erlenmeyer flask. You then add 2-3 drops of phenolphthalein. Trial B: Determining the molarity (M) of acetic acid in vinegar. Trial 1: Using a volumetric pipette, you pipet exactly 5.00mL of the vinegar solution into a 250-mL Erlenmeyer flask. You then add 2-3 drops of phenolphthalein. Trial 2: Using a volumetric pipette, you pipet exactly 5.00mL of the vinegar solution into a 250-mL Erlenmeyer flask.…0.2061 grams of an unknown organic acid was dissolved in about 50 mL of water in a 250 mL Erlenmeyer flask and titrated against 0.1000 M NaOH solution using bromothymol blue indicator. 15.56 mL of titrant was used to achieve end point. Calculate the Neutralization Equivalent. Write answer with two decimal places only. No units please.0.1890 grams of an unknown organic acid containing 2 carboxylic groups was dissolved in about 50 mL of water in a 250 mL Erlenmeyer flask and titrated against 0.1000 M NaOH solution using bromothymol blue indicator. 34.62 mL of titrant was used to achieve end point. Calculate the molar mass of the unknown. Write answer with two decimal places only. No units please.
- Q12 chapter 15 titrations Solve asap hundred percent correct solution neededEach row represents an experimental “run” (in this case, averaged sample of mango wine). Column A shows the run number; Column B shows the percentage of ethanol; Column C shows the amount of glycerol (in g/L); Column D shows the amount of acid (in g/L); Column E shows the temperature of the run (in °C); and Column F shows the pH of the run. Y represents Ethanol of mango wine. 1. Create a scatterplot of the ethanol data for all 20 runs a. Draw a graphical representation of the model that you chose to use. b. Comparing the model to your data, evaluate its fit in one or two sentences. (Not sure what this question is getting at. I've provided all the information given) c. Replace the generic Yi = β0 + ε with the information you have here. Please help. I have no idea what this even means or where to start. HERE is the attached dataset to start with; a b c d e f Run Ethanol Glycerol Acidity Temp pH 1 4.8 3.5 0.84 24 3.8 2 9.6…Stock solution and dilution materials: 500mg ascorbic acid (powdered) and 2L distilled water Solve for: 1. Create a stock solution of 0.042 M 2. Dilute it to 0.012 M answer the ff: 1. amount of ascorbic acid used (in mg) 2. amount of distilled water mixed with the ascorbic acid (in mL)
- 1. A solution contains a mixture of Na2CO3 and NaOH. Using phenolphthalein indicator 25 mL of mixture required 19.5 mL of 0.995 M HCl for the endpoint. With methyl orange, 25 mL of solution required 25 mL of the same HCl for the endpoint. Calculate grams per liter of Na2CO3 in the mixture. a.23b.36 c.78 d.112. A 1.2040 g sample containing sodium carbonate and inert material was dissolved in water and titrated to methyl orange end point, requiring 32.50 mL of 0.1020 M HCl. Calculate the %Na2CO4 in the sample. a. 93.36% b. 14.59% c. 12.75% d. 23.41%3. A 0.1510 g KHP (MW = 204.22 g/mol) sample required a volume of 57.04 mL of NaOH solution to reach a phenolphthalein end point. Calculate the molarity of NaOH titrant. a. 0.02593 M b. 0.006481 M c. 0.01296 M d. 0.01012 MExtraction of Caffeine Post-lab 9. A 12 oz can of Coke contains 46 mg of caffeine. One fluid ounce is equivalent to 29.57 mL(1 oz = 29.57 mL). What is the concentration of caffeine in Coke, in units of mg/mL? Showyour work. 10. What is an alkaloid? 11. Why is anhydrous sodium carbonate used in isolation of Caffeine? What do youthink would happen if we used it during our simulation?What are preservatives? What food products can be consumed with preservatives and additives? What are the adverse effects of consuming food product with food preservatives and additives? pls answer the 3 questions above :>
- A student needs 50.0 mL of 5.74×10 -4 M FD&C Blue Dye No. 1 solution. If the student has 2.00 mM FD&C Blue Dye No. 1 stock solution to work with, how much of the 2.00 mM solution should be added to the 50.0 mL volumetric flask? Hint, 2.00 mM is 0.00200 M. 14.35 mL 0.06971 mL 3.485 mL 12.85 mL 6.971 mLMULTIPLE CHOICE: Choose the letter of the best answer Please answer all questions correctly 1. The standardization of volumetric solution does not require hydrochloric acid. A. Permanganometry B. Cerimetry C. Iodimetry D. lodometry E.All of the choices 2. In the standardization of its volumetric solution, the primary standard is dried to constant weight, which means drying shall be continued until two consecutive weighings do not differ by more than 0.50 mg. A. Permanganometry B. Cerimetry C. Iodimetry D. lodometry E.All of the choices 3. In the standardization of its volumetric solution, potassium iodide was added to A. Permanganometry B. Cerimetry C. Iodimetry D. lodometry E.All of the choices 4. Starch is the indicator of choice for these procedures because it forms intense blue coloration with the solution. A. Permanganometry B. Cerimetry C. Iodimetry D. lodometry E. C and D 5. In the standardization of its volumetric solution, the reaction needs to be…What is the concentration in units of grams of dye per liter of solution ,of the dilute dye solution that is produced as follows? transfer 19.53 ml of stick solution (6.10g/L) to a 200.0Ml volumetric flask and dilute to the 200 ml line with deionized water. Correct number of significant figs

