Q: Below is one of the reactions used in the synthesis of eluxadoline. What type of reaction is it?…
A: Reductive amination
Q: Show how to convert 4-methoxybenzyl chloride to each amine.
A: The given reagent is 4-methoxybenzyl chloride. The product that is to be formed is represented as…
Q: Question 30 What is the best reagent to complete this synthesis? 1) ???? 2) NaCN он CN А. PBR3 В. C.…
A: The correct option is option D.
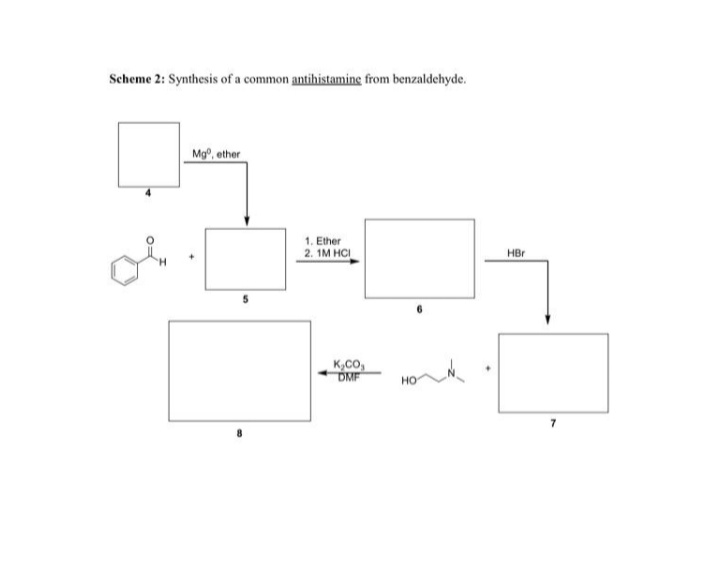
Q: Propose a reagent for the conversion of (B) to (C).
A: This reaction will be a type of condensation reaction as in the reactant side we will be take an…
Q: Why can you not use this reaction to make a secondary amine? a) Alkyl halides are weak…
A: Since in the above reaction, the secondary amine formed will have more inductive effect compared to…
Q: Most Activating 2 Least Activating GOH3 OH Oph CH Pheno? anisure 13r |-naphthol Diphenyl etler…
A: This question is checking the basic concept of nature of Benzylic lone pairs. We have to tackle…
Q: Complete Reaction NBS (N B1omo Succinamide) DCM (CHCI,) dchlore methone
A: Introduction: Addition reaction is a reaction in which substituent get attached to double bond to…
Q: Which reaction is NOT appropriate for the synthesis of 2-pentanone? 1. ВНЗ/ТHF 2. H2O2/NAOH А. of 1.…
A: Solved in step 2.
Q: Choose the most appropriate reagent(s) for conversion of ethyl acetate to acetamide. H OEt OCH2CH3 H…
A:
Q: Nole, formatión of the enolate is carried out under irreversible conditions. 1. LDA, THF, -78 °C…
A:
Q: 21. Give 2 different one-step methods to convert propanoic acid to methyl propanoate.
A:
Q: a. N,N-Dimethyl formamide (DMF) -> 1 Bromo Butane b. 2-methyl-2-butanol -> 2 methyl 2 butene c.…
A:
Q: (-)-Ipc2BOTf i-Pr2NEt ЗА CH2CI2, -20 oC TBSOTF 2,6-lutidine 4B 50 CH2CI2, -78 °C structure of 3A?…
A: The geometry of enolate depends on the chiral borate reagents. (-)Ipc2BOTf majorly produces…
Q: Which of the following will not oxidize benzaldehyde? -- acidified KMnO4 -- acidified K2CrO4 --…
A: Oxidation of aldehyde: When any oxidizing agent in acidic conditions, react with benzaldehyde it…
Q: (4) Can the following alkyd recipe be carried to “complete" conversion without gelling?…
A: Moles f Equivalence (f * mol) pentoerythritol 1.21 mol 4 4.84 phthalic anhydride 0.50 mol 2…
Q: give the structure of phenol plz asap 8:27 am /
A:
Q: A2. Which reagent should be paired with acetic anhydride to produce: ? A. В. C. D. OH OH OH
A:
Q: Select the most suitable set of reagents that can be used in reducing ethanamide CH;CONH2 to…
A: Degradation of amide group to primary amine using Br2 and alkali is called as Hoffmann Bromamide…
Q: d) Write and name a diazonium salt. e) Write and name a 2,3-unsaturated aldehyde molecule. f) Write…
A: Diazonium salt: Its a class of organic compounds having a functional group R−N+2X− where R can be…
Q: Draw the products that result from each step in the synthesis of m-bromoaniline. HNO3, H2SO4 Br2,…
A: Step 1: The reaction of benzene with a mixture of conc. HNO3 and conc. H2SO4 is Nitration reaction (…
Q: . identify the structure of A and B in the following synthetic scheme: Write out complete reactions…
A: By observing the nature of the reagents used, we can expect ketone and imine as products…
Q: Why is alkoxide (RO-) a stronger base than Chloride? Why are alcohols typically poor electrophiles?
A: 1. alkoxide ions are better proton acceptors as compared to hydroxide ion which makes them stronger…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid…
A: The reaction given is,
Q: 46. Which carbonyl group of the given compound is most reactive for nucleophilic addition reaction?…
A: The answer of all questions have been outlined below
Q: Primary amines can also be prepared by the reaction of an alkyl halide with azide ion, followed by…
A:
Q: W1 1. LDA, THF, -78 °C 2. CI CO₂Et RCHO TICI4 (Titanium chelate) W4 W2 W5 LDA THF Br₂, CH₂Cl₂ 0 °C,…
A:
Q: 90. What compound lucidin, rubiadin and munistin are belong? A) chrysacin derivatives B)…
A: A) Compound lucidin , rubiadin , munistin belongs to anthraquinone derivatives. B) Above structure…
Q: `OR OR Benzoate esters can undergo either acid- or base-catalyzed hydrolysis as shown below. HOR +…
A: The reaction is the hydrolysis of benzoate esters.
Q: Proindetie Product CN CN NacN DMSO horcachion
A: Nucleophilic substitution occurs when the leaving group is good.
Q: A very etticient synthesis of resveratrol was reported in 2006. Identify the missing reagents. он…
A:
Q: 1. What is the merchanism of synthesis of salicylamide?
A: Salicylamide: It is a derivative of salicylic acid. It can be synthesized from phenol or directly…
Q: Match the ff reagents: a. Br₂ in CH₂Cl₂ b.CrO₃ , H₂SO₄, acetone c. concentrated HCl with ZnCl₂…
A: Functional groups in organic chemistry refers to specific atomic entities that provide…
Q: Cyclohex-4-ene -1,2-dicarboxylic acid (mp 165°C) A X 4. xylene H20 -SO2 Cyclohex-4-ene…
A: Chelotropic reaction is also a type of pericyclic reaaction. Bond breaking and making occurs…
Q: AICI3 K Zn(Hg) HCI , L para product Toluene Succinic anhydride SOCI2 H2SO4 NABH4 AICI3 N M heat P.
A: The aromatic compound gives an electrophilic substitution reaction. Toluene gives Friedel-Krafr…
Q: How would you convert 1-chloro-3-methylbutane to N-ethyl-4-methylpentanamine. a) NH₂CH₂CH3, DCC b)…
A: In the given reaction the 1-chloro-3-methylbutane is converted into N-ethyl-4-methylpentanamine by…
Q: Match each item to a choice: Choices: alkene Reductive amination Curtius rearrangement Hoffman…
A: Match the following of amine reactions
Q: Devise a 5-step synthesis of the carboxylic acid from acetylene. 1. reagent 1 2. reagent 2 3.…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the amine side product. N'…
A: Given reaction is : Draw the major product of the reaction = ?
Q: Select the single best answer. What reagents are needed to convert phenylacetylene into acetophenone…
A: Answer:- The alkynes on hydration in the presence of sulfuric acid and HgSO4 , gives the…
Q: Activating 3. Least Activating GOH3 HIM CH3 Pheno? 13r 1-naphthol anisue Aceteniite Diphenyl etler…
A: Solution : An activating group is a group that increases the electron density on the ring like…
Q: 10.₁ amide? A. one equivalent B. two equivalents C. this conversion is not possible D. one…
A: Following questions are belongs to the reaction mechanism.
Q: Provide the missing reagents for each of the following sreactions: CH2 но он CH NH2 но DFocus ENG…
A:
Q: To phenylmagnesium bromide was added b) ethyl benzoate. c) benzoylchloride d) benzaldehyde e)…
A: This is reaction of grignard reagent with functional group. Acid derivatives functional groups need…
Q: Among the following approved sweeteners, which has the highest relative sweetness? a)…
A: A multiple choice question based on general chemistry that is to be accomplished.
Q: (d). 1. MgBr 1. O3 (2 equivalents) 2. Me,s 2. H20 K2Cr,07 H2SO4 (2 equivalents) (d) Propose an…
A:
Q: Describe what each reagent does in SDS Page 1. Sodiun dodecyl sulfate 2. Polyacrylamide 3.…
A: The estimation of protein is nowadays carried out by SDS-PAGE methodology. It is an electrophoresis…
Q: the Curven Compound is a SH N best design Strategy Explain N LOCAD fontiometabolitics (8) cell cycle…
A: The given molecule is a heterocyclic compound with the chemical formula C5H4N4S.
Q: How many grams of adapalene 0.1 % (w/w) gel is required to make 90 grams of adapalene 0.025 % (w/w)…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).SHOW COMPLETE SOLUTIONS Q3. Which isolate has A260/280 ratio of above 2.0?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…Show how the synthetic scheme developed in Problem 23.67 can be modified to synthesize this triiodobenzoic acid X-ray contrast agent.How to approach and solve the problem, thank u

