Section 1: Phase Diagrams nics 2.5 1. Label the triple point on your phase diagram. 2. Label the critical point on your phase diagram. 3. Label the solid, liquid and gas sections on your phase diagram. 4. At what pressure should freezing points and boiling points 1.5 be read? 5. What is the freezing point of this substance? 6. What is the boiling point of this substance? 7. Beginning at 1.5 atm and 50 K, what phase change takes place when this substance heats to 200 K at a constant pressure? 8. Beginning at 200 K and 2 atm, what phase change takes place when this substance drops to 0.2 atm at a constant temperature? 9. Does this phase diagram represent water? Explain your 0.5 100 200 300 400 500 Temperature (K) answer. Pressure (atm)
Section 1: Phase Diagrams nics 2.5 1. Label the triple point on your phase diagram. 2. Label the critical point on your phase diagram. 3. Label the solid, liquid and gas sections on your phase diagram. 4. At what pressure should freezing points and boiling points 1.5 be read? 5. What is the freezing point of this substance? 6. What is the boiling point of this substance? 7. Beginning at 1.5 atm and 50 K, what phase change takes place when this substance heats to 200 K at a constant pressure? 8. Beginning at 200 K and 2 atm, what phase change takes place when this substance drops to 0.2 atm at a constant temperature? 9. Does this phase diagram represent water? Explain your 0.5 100 200 300 400 500 Temperature (K) answer. Pressure (atm)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter9: Liquids And Solids
Section: Chapter Questions
Problem 17QAP
Related questions
Question
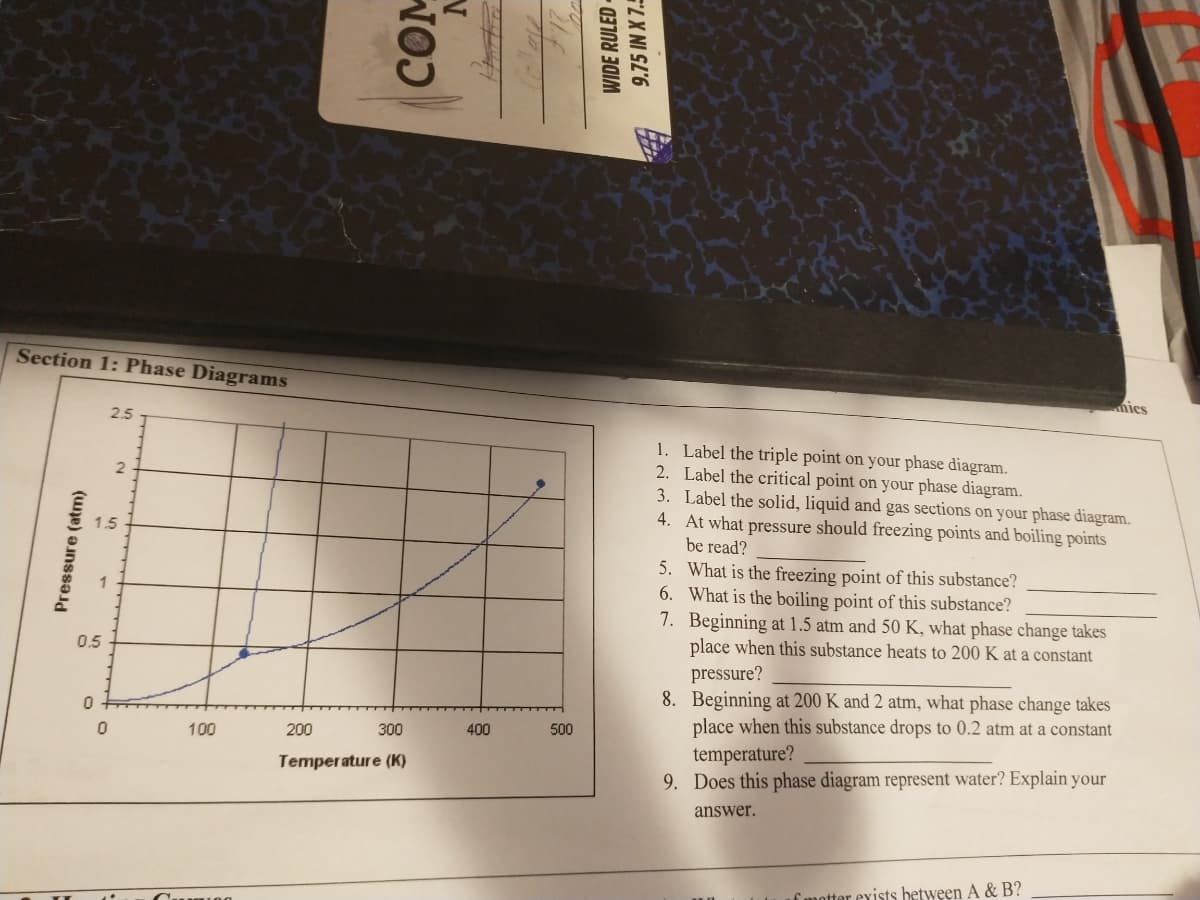
Transcribed Image Text:Section 1: Phase Diagrams
nics
2.5
1. Label the triple point on your phase diagram.
2. Label the critical point on your phase diagram.
3. Label the solid, liquid and gas sections on your phase diagram.
4. At what pressure should freezing points and boiling points
be read?
1.5
5. What is the freezing point of this substance?
6. What is the boiling point of this substance?
7. Beginning at 1.5 atm and 50 K, what phase change takes
place when this substance heats to 200 K at a constant
0.5
pressure?
8. Beginning at 200 K and 2 atm, what phase change takes
place when this substance drops to 0.2 atm at a constant
temperature?
9. Does this phase diagram represent water? Explain your
100
200
300
400
500
Temperature (K)
answer.
i motter exists between A & B?
Pressure (atm)
COM
WIDE RULED -
9.75 IN X 7.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning