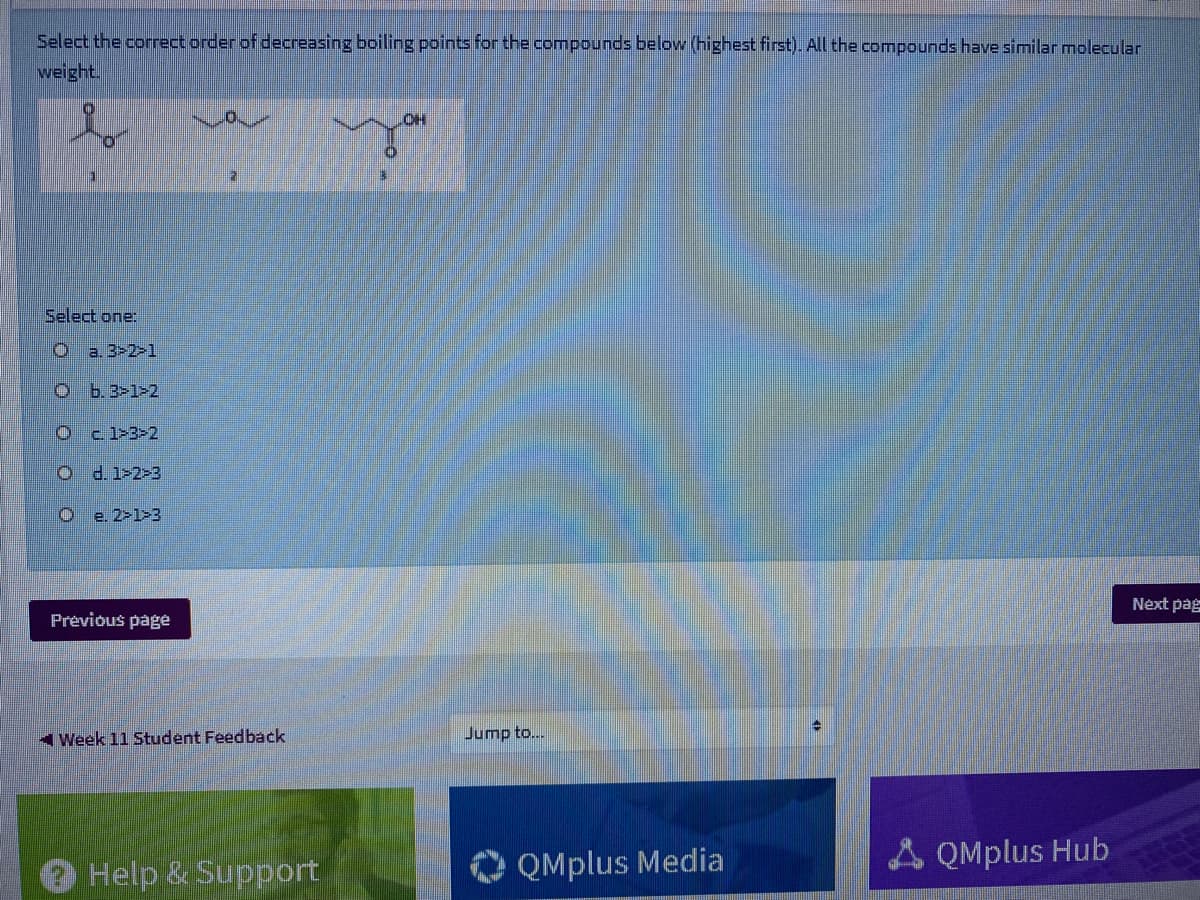
Select the correct order of decreasing boiling points for the compounds below (highest first). All the compounds have similar molecular weight. OH
Q: At what temperature is cyclohexane melting?and why does it melt there
A: Melting temperature of cyclohexane is 6.47°C It have large surface area so slightly have high…
Q: Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with…
A: A hydrogen bond is formed by the atoms fluorine,oxygen and nitrogen with hydrogen atom.
Q: Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with…
A: Hydrogen bond is the bond that is formed between the small atom and the electronegative atom.
Q: Circle the molecule that has a higher boiling point in each pair. (b) i VS. (c) VS. VS.
A:
Q: Which of the following compounds is expected to have the LOWEST boiling point? a. CH3OCH3 b.…
A: Solution The temperature at that liquid force per unit area equals air pressure is spoken as boiling…
Q: А) ОН B) LOH ОН C)
A: Melting point of a substance is defined as the temperature at which the substance changes its phase…
Q: Rank the following molecules in approximate order of melting temperature, from the lowest melting…
A: The melting point depends on the force of attraction between the molecules. More is the force of…
Q: А. В. E. (А) A (B) В (C) С (D) D (E) Е
A: Boiling point depends on 1) molecular weight 2) cis or trans arrangement 3) branching
Q: What is the correct IUPAC name for the compound shown here? он OH
A:
Q: which compound has the highest boiling point: hexan-2-on or hexan-2-ol?
A: The structures of the given compounds, hexan-2-on or hexan-2-ol are as shown below.
Q: Which compound in the following pairs will have the higher boiling point? Explain your reasoning 1.…
A: Given We know about the boiling point of following compound 1) NH3 and PH3 2) ethylene glycol and…
Q: Arrange these compounds in order of increasing boiling point.Explain your reasoning.a. CH4 b. CH3CH3…
A: Rules : 1) When the molecular mass of molecules are too different (i.e not approximately same) than…
Q: Arrange the following compounds in order of increasing their boiling points (weakest to strongest).…
A: Given here some compounds and we are asked to arrange the givencompounds in order of their boiling…
Q: Explain why CH3CH2NHCH3 has a higher boiling point than (CH3)3N, even though they have the same…
A: Hydrogen bonding is a special type of dipole-dipole interaction in a polar bond which has hydrogen…
Q: Which member of each pair has the lower boiling point? Explain your choice. ***When you draw the…
A: Boiling point depends on surface area ( Branching increases boiling point decreases). Higher surface…
Q: Clear All CH;CH,C-H Lowest boiling point CH,CH2CH2CH3 Intermediate boiling point Highest boiling…
A:
Q: Which of the following pure compounds can form hydrogen bonds? Which can form hydrogen bonds with…
A: Hydrogen bonding- The interaction between electropositive ‘H’ atom of first molecule and…
Q: Which of the molecules below has the higher boiling point? Briefly explain your choice.…
A:
Q: Which of the following substances would be expected to have the lowest melting point ? A)…
A: As we know that melting point is directly proportional to the molecular mass. Here molecule D( CH4)…
Q: arrange the following compounds in increasing boiling point a. CH3CH2CH2CH2OH b. CH3CH2OH c.…
A: The boiling point of a compound depends primarily on the intermolecular forces of attraction. The…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: General chemistry
Q: What is the correct IUPAC name for the compound shown here? OH
A: In the IUPAC system of nomenclature, the longest carbon chain containing the hydroxyl group is…
Q: Which has a higher boiling point and indicates the most important reason. Circle the compound with…
A: KBr is an ionic compound as opposed to the molecular compound CH3CH2 As ionic interactions are…
Q: Arrange the following compounds in order of increasing boiling point: Increasing of boiling point…
A: Welcome to bartleby ! Introduction : We have to arrange boiling point .
Q: The above compound has a boiling point than pertan-1-of because it lacks
A: Hydrogen bonding: it can also be defined as the electrostatic force of attraction between hydrogen…
Q: IV. Which member of each pair has the higher boiling point? Explain briefly your answers. 1.…
A:
Q: Why CH3CH2NHCH3 has higher boiling point tha (CH3)3N even they have the same molecular weight
A: First compound CH3CH2NHCH3 is secondary amine, ethyl methyl amine while second Compound (CH3)3N is…
Q: Which member in each pair of substances has the higher boiling point? Explain. a) NH3 or PH3 b)…
A: Boiling point is the temperature at which the liquid state of a compound converts to vapor state of…
Q: Rank the following compounds in order of decreasing boiling point. Question List (6 items) (Drag and…
A: In the given question there are six alkanes, we have to rank boiling point from higher to lower.…
Q: The compounds in each part below have the same (or similar) molecular weights. Which compound in…
A: Boiling point of an organic compounds depends directly on the length of a Carbon chain, ie. longer…
Q: Arrange the following sets of compounds in order of their decreasing boiling points. CH3(CH2)40H,…
A: The compounds given belong to same homologous series A series of organic compounds having a…
Q: Arrange the compounds from highest boiling point to lowest boiling point. Highest boiling point…
A:
Q: For each of the three horizontal series below, rank each molecule from highest (1) to lowest (4)…
A: Answer Series 1: Generally boiling point of alcohols are increase with increasing molecular mass, In…
Q: Directions: In each item, rank the given compounds according to the given description. Write only…
A:
Q: pent-3-ene nylhex-3-ene nylhex-3-ene
A: 1- Firstly selected the long chain carbon bone. 2- give the numbering on long chain carbon. 3-…
Q: Rank each set of compounds in order of increasing boiling points.ethanol, dimethylamine, dimethyl…
A: Boiling point : It is the temperature in which liquid will turn into vapor. Boiling point depends…
Q: 0. Why does ethanol have a higher boiling point than dimethyl ether despite having the same…
A:
Q: Which compound has the lowest boiling point? CH3 CH3 CHCH2CH3 CH. CH;CH,CH,CH,CH3 CH3-C-CH3 ČH3 II…
A:
Q: Pick the compound with the highest boiling point in each pair.Explain your reasoning.a. CH3OH or…
A: boiling point - Which temperature the liquid convert into gas is called boiling point of compound .…
Q: In each pair of compounds, pick the one with the higher boiling point. Explain your reasoning.a.…
A: a) In the given compounds methanol has the hydrogen bonding interactions along with dispersion…
Q: HHH HH H-C-C-C-c-C-H Rank these compounds from highest to lowest boiling point. H-C-H pe H-C-C-C-H…
A:
Q: Which has the LEAST negative heat of combustion among the compounds below? Select one: a. "tBu Ob.…
A: Amount of heat need to completely burn the one mole of compound is known as heat of combustion of…
Q: Which of the following compounds will have the highest boiling point? O CH3CH(CH3)CH3 O CH3CH2CH3…
A: Compound in which hydrogen bonding present will have higher boiling point.
Q: Pick the compound with the highest vapor pressure at a given temperature. Group of answer choices…
A: Vapor pressure is defined as the pressure exerted by the vapor in the thermodynamic equilibrium. The…
Q: Arrange the following sets of compounds in order of their decreasing boiling points. A. CH3(CH2)4OH,…
A:
Q: CH3ch2ch2ch2ch2Oh Boiling-oing lowest, intermediate, highest boiling point…
A:
Q: 4. One of the compounds shown has a boiling point of 37 °C and the other has a boiling point of 60…
A: Dipole moment- It measures the polarity of a chemical bond within a molecule. It occurs whenever…
Q: Rank the following by increasing boiling points, so that 1 is the highest and 4 is the lowest.…
A:
Q: Which molecule do you expect to have the lowest boiling point? OCH3CH2CH3 CH3CH2CH2CH2CH3 O…
A: Interpretation: We have to tell which of the following has lowest boiling point.
Q: Which has higher boiling point? A. CH3CH2CH2CH2CH3 B. (CH3)2CHCH2CH3
A: The boiling point of pentane is 36.1oC. The boiling point of isopentane is 27.8oC. Hence, the…
Step by step
Solved in 3 steps with 3 images

- 1. Which of the following compounds can form hydrogen bonds with water?a. octaneb. HClc. NaCld. acetic acid2. Maria wants to determine the percent water content of an unknown hydrate of AB. Given the following data:mass of empty crucible - 12. 2784gmass of AB · xH20 + crucible - 17.4960gmass of AB + crucible - 15.1314gCalculate the % water content of the hydrate3. From the above data, give the correct formula for the hydrate. (AB₂ Molar mass = 129.84 g/mol)Identify whether the statements are TRUE or FALSE 1. Mohr pipettes have marks always end before the tip. 2. Serological pipettes have marks that continue all the way down to the tip.If Compound Y is characterized by having a melting point of 124°C under normal atmospheric pressure. Determine which of the following samples (VWXY) is high likely to contain the highest % of Compound Y? Sample W (114-122°C) Sample V (126-131°C) Sample Y (121-123°C Sample X (119-120°C)
- 1. The mixture below is seperated using HPLC with a octadecylsiloxane (nonpolar) stationary phase and a methonal (CH3OH)/water mobile phase. In what ofder will the compnents come off the column. What would happen to the order in which the components come off a polar colum if pentane is used as the mobile phase? Heptane, Heptanol, Butyl propyl etherCH.8 #8 - Please fill in the blank, cirlce your answer for thumbs up!Plz label the following
- Melting point: 98-105 Percent yield: 60% did you successfully make pure product? Explain.Given the information listed below, identify the compound. Physical description: This substance is a colorless but fragrant liquid that is oily in appearance. Chemical information: Observed Melting point: -78.5 °C Observed Boiling point: 138-142 °C Calculated MW: 130 g/mol Density: 0.876 g/cm3 Solubility in water: Minimally soluble, Other: Flash Point: 77 °F Spectroscopic information: IR bands indicate this compound contains C-H functionalities and the appearance of a strong stretch near 1700 cm-1 as well as several strong bands between 1500 and 1000 cm-1. The C-13 NMR spectrum for this compound indicates 6 unique carbon atoms and no cyclic structure. The proton NMR data indicate 5 sets of unique protons. C-13 NMR data of : PPM: 171.08, 63.13, 37.48, 25.18, 22.51, 20.96 Int: 222, 887, 645, 635, 1000, 473 Assign: 1, 2, 3, 4, 5, 6 H-1 NMR Assign: A, B, C, D, E Shift: 4.095, 2.037, 1.693, 1.521, 0.9261. Write three major criteria of forming a Hydrogen bond in bullet points with example. 2. Certain amount of pure charcoal (carbon) has to be burnt with supplied natural air. Process engineer allowed the exact amount of air required to burn the entire charcoal completely to carbon dioxide. Carbon has no reaction with nitrogen gas present in the air. In the reaction outlet if 5 kg of nitrogen gas was found, calculate the amount of charcoal used. (12C, 16O, 14N)
- Give me handwritten answer plz?C. PARAFFIN1. Place a paraffin about the size of a rice grain in a dry test tube. Warm gently andadd e drops of bromine (CAUTION!). Result: ____________________________________2. Melt a small piece of paraffin in a dry test tube and add 3 drops of potassiumpermanganate solution. Result: _______________________________________________What does this prove? ________________________________________________________D. LPG (Liquefied Petroleum Gas)CAUTION: No flame must be lighted in this portion.Attach a glass nozzle to a rubber tubing and connect this to the gas outlet.Prepare 2 test tubes containing 1 mL of each alkaline potassium permanganateand bromine water. Slowly open the gas cock and bubble the gas for one minuteinto each tube. Property of and for the exclusive use of SLU. Reproduction, storing in a retrieval system, distributing, uploading or posting online, or transmitting in any form or by anymeans, electronic, mechanical, photocopying, recording, or otherwise of any part of this…How to convert umol CH4 m-2 d-1 to mg CH4 m-2 d-1 and vice versa? Thank you.



