Q: 2) Provide the missing information in the following sequences. Pd /H2 HF Starting Material Product…
A:
Q: What is the major organic product? 1) EILI (excess) Et20 2) H20 C OH но
A: EtLi is a type of organolithium compound. Where, the organo part is an alkyl group , R (Here, R=Et).…
Q: Select the primary reaction product
A: The given reactant is 5-hydroxypentanoic acid.
Q: Draw the product(s) for the following reaction. Upload your images as one file on the last question.…
A: Answer:- this question is answered by using the simple concept of hydrogenation of unsaturated fats…
Q: This question has multiple parts. Work all the parts to get the most points. For the reaction below:…
A:
Q: Complete the following reaction by drawing structural formulas for the organic product(s). CH OCH +…
A:
Q: When a reaction is desired between two incompatible reagents, what is useful to employ? A Protecting…
A: A protecting group is a molecular framework that is introduced in a reaction to block the reactivity…
Q: NH₂ CI 1. Acyl Chloride and AICI, catalyst 2. Zn, Hg, HCI Cl₂ and FeCl3 catalyst HNO3 and H₂SO4 1.…
A: Here we have to predict the major product formed in the following given reactions. Here elctro…
Q: uv CH;CH;CH;CH3 + Br2 A + B State the type of reaction. Nyatakan jenis tindak balas tersebut. What…
A:
Q: Table 6.6 Observations on the reaction of each test sample with ammoniacal silver nitrate. Sample…
A: Tollence reagent that is ammonical silver nitrate , that acts as weak base . Thus strongly acidic…
Q: НООС OH 1 2 H2 1. p-Toluenesulfonic acid Oppenauer Oxidation HO Lindlar Catalyst 2. H2, Pd/C
A: For the above organic synthesis, the following transformations must be known: 1. H2 in presence of…
Q: Table 6.5 Observations on the reaction of each test sample with sulfuric acid. Sample Observations…
A: Sulfuric acid solubility testThe T1 electrons of alkenes and alkynes are very reactivetowards…
Q: NH₂ CI 1. Acyl Chloride and AICI, catalyst 2. Zn, Hg, HCI Cl₂ and FeCl3 catalyst HNO3 and H₂SO4 1.…
A:
Q: CH2 = CH2 + Br+ CH2-CH2Br This is a step in a ____ reaction where ____ is the electrophile
A: The given reaction is, CH2=CH2 + Br ---> CH2-CH2Br The given reaction is an electrophilic…
Q: Step 8 HO. HN. HN
A:
Q: What organic product is produced in the following reaction? CH2=CH2 H, H20 A) HOCH2CH(OH)2 B)…
A: The solution is as follows :
Q: В. a. technical/commercial reagents aibr b. U.S.P and N.F. reagents C. chemically pure reagents d.…
A: Grades for Chemicals and ReagentsIt is imperative that everyone in the custody supply chain know and…
Q: Identify the functional groups in each molecule.
A: A functional group is a specific group of atoms or bonds within a compound that is responsible for…
Q: Provide the term that matches the definition. Definition: transfer of energy from the system to the…
A: Provide the term that matches the definition. Definition: Transfer of energy from the system to the…
Q: 2. Br HCI, H,O 1. Cl, NaOH, H,0 2. H. H,0 1. P. Br 2. H,0 он 3. CH,NH, 1. LDA EIONA (2 equiv.)…
A: o LDA is a bulky base that abstracts proton from the less hindered site o Under haloform reaction…
Q: 1. A. Fill in the structure of the products in the box below. HCI, H2O B. Name the reactant above.
A: The reaction is as follows,
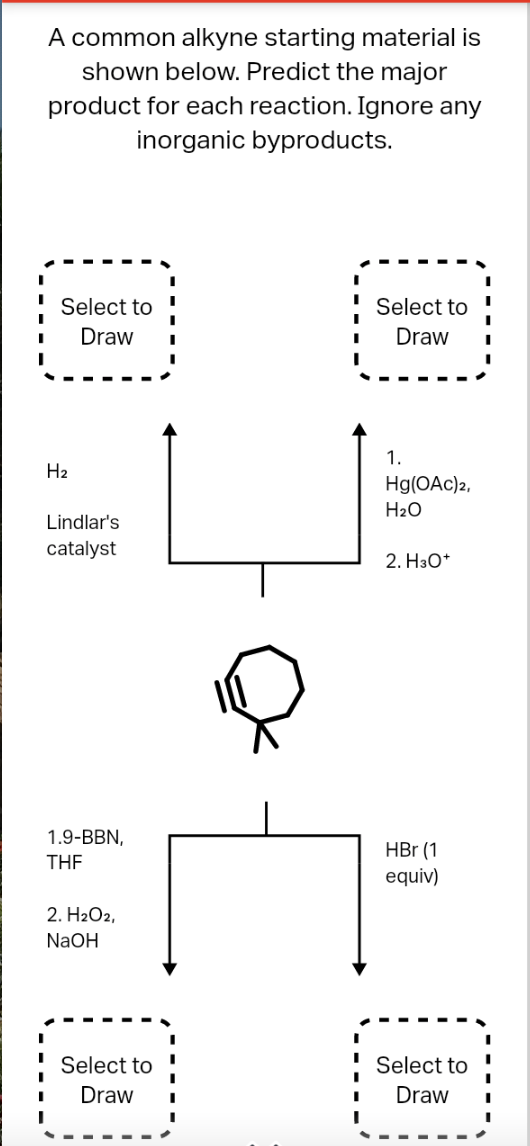
Q: Select to Draw Cl2 Select to Draw OH SOCI2 pyridine H₂ Lindlar's catalyst Select to Draw CH3CCNa THF…
A:
Q: What is the reagent for the following reaction? Na in liquid ammonia H2, Lindlar's catalyst…
A:
Q: Provide the missing reagent for the below reaction: H. CH H,C H,C CH CH O H Pd/C Na NH H Lindlar's…
A: Correct answer is H2 ,Lindlar's catalyst. Alkyne is hydrogenated in presence ofLindar's catalyst to…
Q: QUESTION 9 Choose the best reagent for the following reaction. OH .CI ? SOCI2, pyridine HCI Cl2…
A: Here, the given alcohol is secondary (2°) and in the product, inversion happens. That means the…
Q: Cresol red Thymel bue Erytvosin B 24Dinitrophenol Bromphenol be Methyl orange Bromcresol green…
A: The question is based on the concept of titration. we are titrating a strong acid with a strong. we…
Q: Considering the following reaction, draw the structures of A and B in the sketchpad. HO. ELOH /…
A:
Q: Draw structural formulas for organic products A and B in the window below. Cul H₂C=CHBr Li pentane A…
A: When alkylhalide is treated with Lithium (Li) it gives an organometallic compound alkyl lithium…
Q: Draw a structural formula for the major product of the acid-base reaction shown. H2N H N. HCI (1…
A: According to Lewis acid-base concept, the base is a substance that can furnish the electrons to…
Q: Draw structural formulas for organic products A and B in the window below. CH3 CH3CH₂CHCHCH3 Br Li…
A: Given reactions are a prepration scheme of gilmans reagent.
Q: h Consider this reaction when answering the following question(s): :0: :0: :0: acid H₂C CH3 CH3…
A: Organic compounds can be defined as the compounds that contain carbon and hydrogen atoms in a…
Q: OH
A:
Q: Select to Draw 1. NaNHa 2. CHCH₂Br Please select a drawing or reagent from the question area
A: Kindly repost your question seperately with clear image.
Q: CH3 1.BH3/THF a) 2. H2O2/-OH
A: The given reactions involve various organic reactions like hydroboration-oxidation, reduction,…
Q: Method of separation/chemical reaction of 3kg sodium borate and 1.5kg hydrochloric acid
A:
Q: Draw structural formulas for organic products A and B in the window belOW. ÇI Li CH,OH CH3CHCH3 A…
A:
Q: Draw the product of benzene and Cl2/FeCl3
A: Given Reaction of benzene with Cl2 in the presence of iron catalyst FeCl3
Q: Draw structural formulas for organic products A and B in the window below. CH3 CH3CCH₂CI _ CH3 Mg…
A:
Q: NO2 KMNO4 H2O
A: When an aromatic compound with an alkyl side chain is oxidized with potassium per magnet, it is…
Q: Need help with e,d,g
A:
Q: What is the major organic product? 1) EtLi (excess) Et20 2) H20 А. Но OH D. B. A.
A: We have to determine the product formed of the following reaction :
Q: Test Oxidation with K2Cr2O7 Lucas Test DNP test Formation of a greenish solution and Cloudy Result…
A: Primary alcohols can be oxidized to aldehydes and carboxylic acids. Secondary alcohols can be…
Q: 1. How many types reaction only in organic chemistry. O A. Seven. OB. Five. O C. Eight.
A:
Q: Which reagent could be uscd to distinguish between CH,COCH, and CI,CHO? A 2,4-Dinitrophenylhydrazine…
A: Answer :- (B) AgNO3/NH3(aq) AgNO3/NH3(aq) reagent is used to distinguish between CH3COCH3 and…
Q: Select a reagent to separate the following compounds (one reagent only) (explain clearly what…
A: When two or more compounds combined chemically to form an insoluble salt, this reaction is called…
Q: Alkyl bromide B 1. diethyl malonate NaOEt, EtOH 2. NaOH, H2O 3. H30*, heat Br `OH
A: This is an example of synthesis of carboxylic acid from diethyl malonate
Q: Draw the structure of the major organic product(s) of the reaction. H3C 1. DIBAH, toluene 2. H30*…
A: Applying concept of organic synthesis.
Q: How can you monitor the progress of a Grignard reaction? O Use TLC to monitor the concentration of…
A:
Q: What needs to be done to make the following reaction go to starting materials? CH;CH,NH2 NCH2CH3…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-ynei need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Given this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…Provide the E2 mechanism for β-elimination reactionemploying 2-chloro-2-methylbutane to prepare 2-methyl-2-butene and 2-methyl-1-butene reaction. Use the actual structures of thereactants and products. Explain which of the alkenes is the major product ofthis reaction ?
- please provide the machanisms of 1a, 1e, 1f1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveI have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:
- Predict the major organic product formed when the compund shown below undergoes a reaction with H2O2 and then is heated in H2O. The 2nd photo is what I thought was the answer, but when I put it into Mastering Chemistry, it was counted as wrong and my feedback was, "What configuration is the C=C bond expected to haev?" I'm confused as to what this is referring to, so I appreciate any help given.Disiamylborane adds only once to alkynes by virtue of its two bulky secondary isoamylgroups. Disiamylborane is prepared by the reaction of BH3 # THF with an alkene.(a) Draw the structural formulas of the reagents and the products in the preparation ofdisiamylborane.(b) Explain why the reaction in part (a) goes only as far as the dialkylborane. Why isSia3B not formed?Help with the following ochem reaction scheme... Consider 3,4-dimethylpiperidine being subjected to the below reaction scheme: step 1) CH3I (excess) step 2) NaOH, heat step 3) CH3I (excess) step 4) NaOH, heat Provide the bond line structures for the major organic product obtained in each step and then discuss the regiochemistry for step 2.

