Selected bond dissociation enthalpies are given in this table. is made by addition polymerization of vinyl chloride (C2 H3 Cl). Vinyl chloride is synthesized from ethylene (C2H4) in a two-step process involving the following equilibria: Dissociation enthalpy (kJ/mol) Bond • Equilibrium 1: C2 H4(g) + Cl2 (g) = C2H4C12 (g) Equilibrium 2: C2 H4 Cl2 (g) = C2 H3 Cl(g) + HC1(g) C-C 348 C=C 614 C=C 839 Part A C-C1 328 С-Н 413 What are the C-C bond orders in C2 H4 Cl2 and C2H3 Cl, respectively? Cl–Cl 242 Express your answers numerically separated by commas. H-H 436 • View Available Hint(s) H-C1 431
Selected bond dissociation enthalpies are given in this table. is made by addition polymerization of vinyl chloride (C2 H3 Cl). Vinyl chloride is synthesized from ethylene (C2H4) in a two-step process involving the following equilibria: Dissociation enthalpy (kJ/mol) Bond • Equilibrium 1: C2 H4(g) + Cl2 (g) = C2H4C12 (g) Equilibrium 2: C2 H4 Cl2 (g) = C2 H3 Cl(g) + HC1(g) C-C 348 C=C 614 C=C 839 Part A C-C1 328 С-Н 413 What are the C-C bond orders in C2 H4 Cl2 and C2H3 Cl, respectively? Cl–Cl 242 Express your answers numerically separated by commas. H-H 436 • View Available Hint(s) H-C1 431
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter16: Spontaneity Of Reaction
Section: Chapter Questions
Problem 78QAP
Related questions
Question
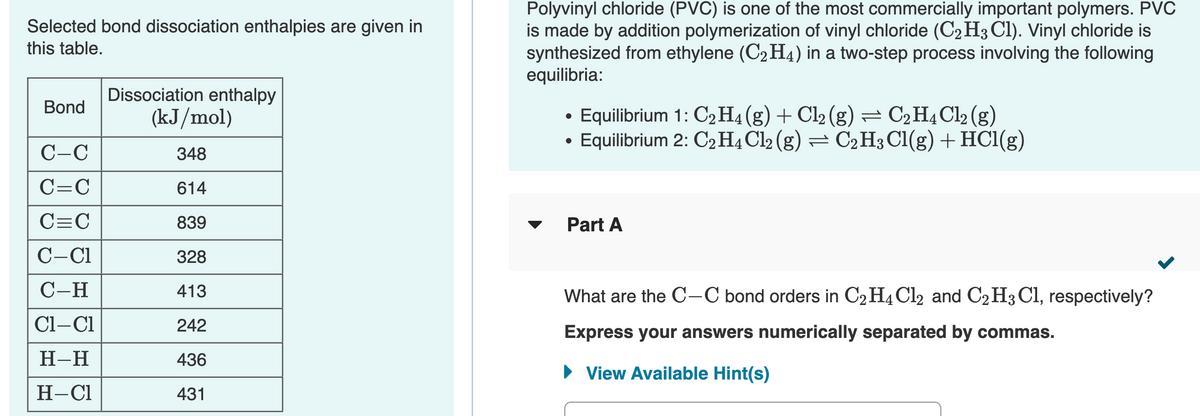
Transcribed Image Text:Polyvinyl chloride (PVC) is one of the most commercially important polymers. PVC
is made by addition polymerization of vinyl chloride (C2H3 Cl). Vinyl chloride is
synthesized from ethylene (C2 H4) in a two-step process involving the following
equilibria:
Selected bond dissociation enthalpies are given in
this table.
Dissociation enthalpy
Bond
Equilibrium 1: C2H4(g) + Cl2 (g) = C2H¾C12 (g)
Equilibrium 2: C2H4 Cl2 (g) = C2H3 Cl(g) + HC1(g)
(kJ/mol)
C-C
348
C=C
614
C=C
839
Part A
C-C1
328
C-H
413
What are the C-C bond orders in C2H4C12 and C2H3 Cl, respectively?
Cl-Cl
242
Express your answers numerically separated by commas.
Н-Н
436
• View Available Hint(s)
Н-СІ
431
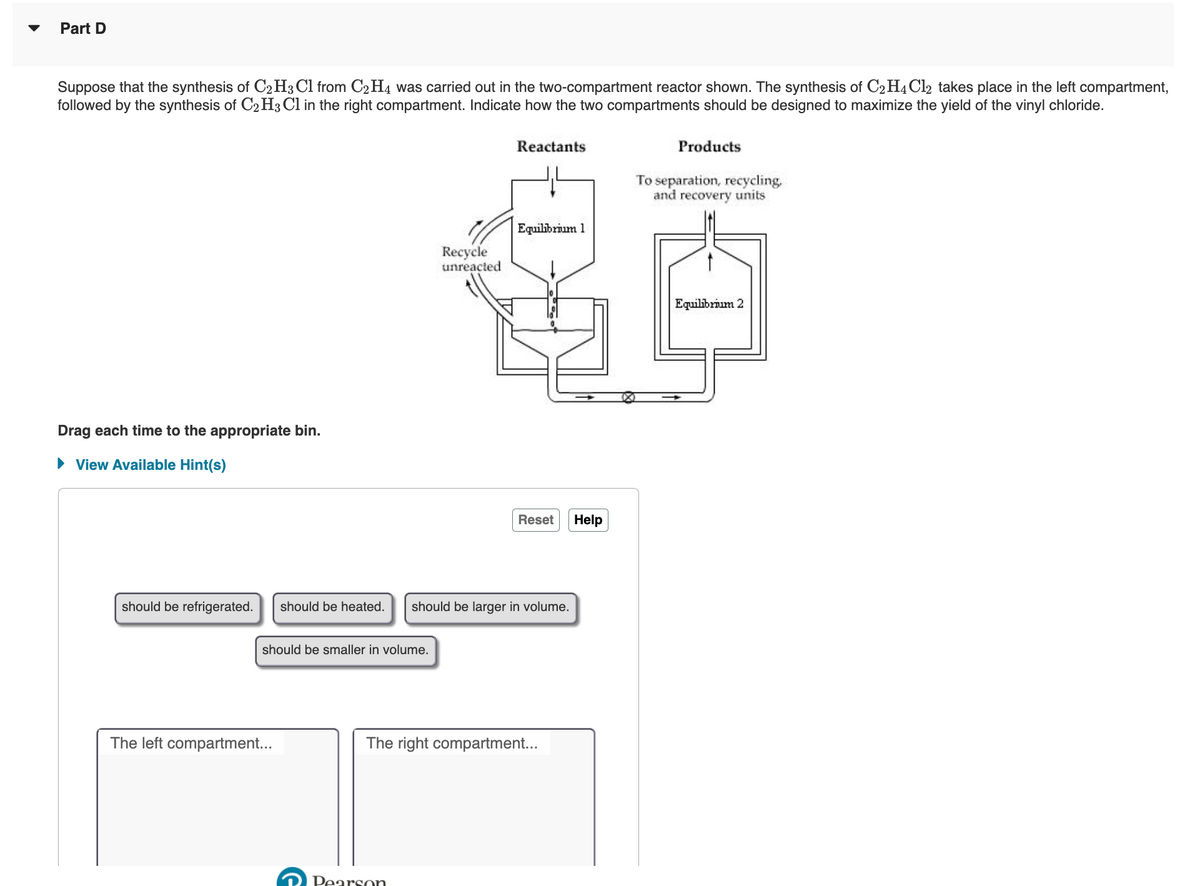
Transcribed Image Text:Part D
Suppose that the synthesis of C2H3 Cl from C2H4 was carried out in the two-compartment reactor shown. The synthesis of C2H4Cl2 takes place in the left compartment,
followed by the synthesis of C2 H3 Cl in the right compartment. Indicate how the two compartments should be designed to maximize the yield of the vinyl chloride.
Reactants
Products
To separation, recycling,
and recovery units
Equilibrium 1
Recycle
unreacted
Equilibrium 2
Drag each time to the appropriate bin.
• View Available Hint(s)
Reset
Help
should be refrigerated.
should be heated.
should be larger in volume.
should be smaller in volume.
The left compartment...
The right compartment..
Pearson
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning