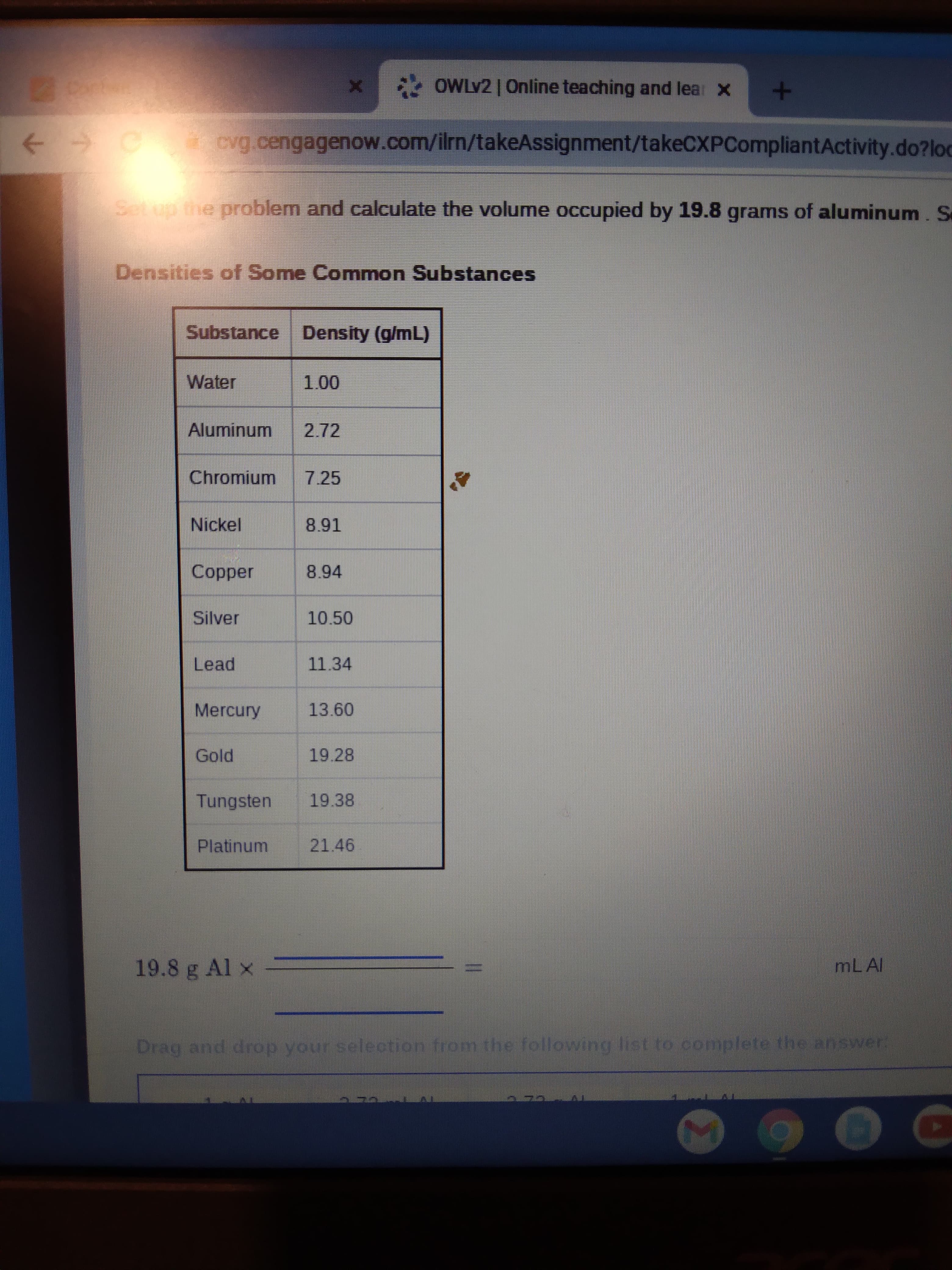
Setup the problem and calculate the volume occupied by 19.8 grams of aluminum. Densities of Some Common Substances Substance Density (g/mL) Water 1.00 Aluminum 2.72
Setup the problem and calculate the volume occupied by 19.8 grams of aluminum. Densities of Some Common Substances Substance Density (g/mL) Water 1.00 Aluminum 2.72
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Transcribed Image Text:EGO
* OWLV2 | Online teaching and lear x
cvg.cengagenow.com/ilrn/takeAssignment/takeCXPCompliantActivity.do?loc
Setup the problem and calculate the volume occupied by 19.8 grams of aluminum. Se
Densities of Some Common Substances
Substance Density (g/mL)
Water
Aluminum
2.72
Chromium
7.25
Nickel
Copper
8.94
Silver
10.50
Lead
11.34
Mercury
13.60
Gold
19.28
Tungsten
19.38
Platinum
21.46
19.8 g Al x
MLAI
Drag and drop your selection from the following list to complete the answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning