52% (4) File Edit View History Bookmarks People Tab Window Help Wed 1:31 PM Chrome Isa * Mind Tap - Cengage Learning Answered: Q Search this coursR G zncl2 molar mass - Google Sear G screenshot on macbook - Google x X A ng.cengage.com/static/nb/ui/evo/index.html?elSBN=9781305657571&id=658288198&snapshotld=1494736& Tutored Practice Problem 3.4.3 COUNTS TOWARDS GRADE Close Problem Calculate percent yield. Consider the reaction of C,H, with O, to form CO2 and H2O. If 3.53 g O2 is reacted with excess C,H6 and 3.62 g of CO2 is ultimately isolated, what is the percent yield for the reaction? Percent yield = Check & Submit Answer Show Approach FEB1 étv 12 POF
52% (4) File Edit View History Bookmarks People Tab Window Help Wed 1:31 PM Chrome Isa * Mind Tap - Cengage Learning Answered: Q Search this coursR G zncl2 molar mass - Google Sear G screenshot on macbook - Google x X A ng.cengage.com/static/nb/ui/evo/index.html?elSBN=9781305657571&id=658288198&snapshotld=1494736& Tutored Practice Problem 3.4.3 COUNTS TOWARDS GRADE Close Problem Calculate percent yield. Consider the reaction of C,H, with O, to form CO2 and H2O. If 3.53 g O2 is reacted with excess C,H6 and 3.62 g of CO2 is ultimately isolated, what is the percent yield for the reaction? Percent yield = Check & Submit Answer Show Approach FEB1 étv 12 POF
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.12QAP
Related questions
Question
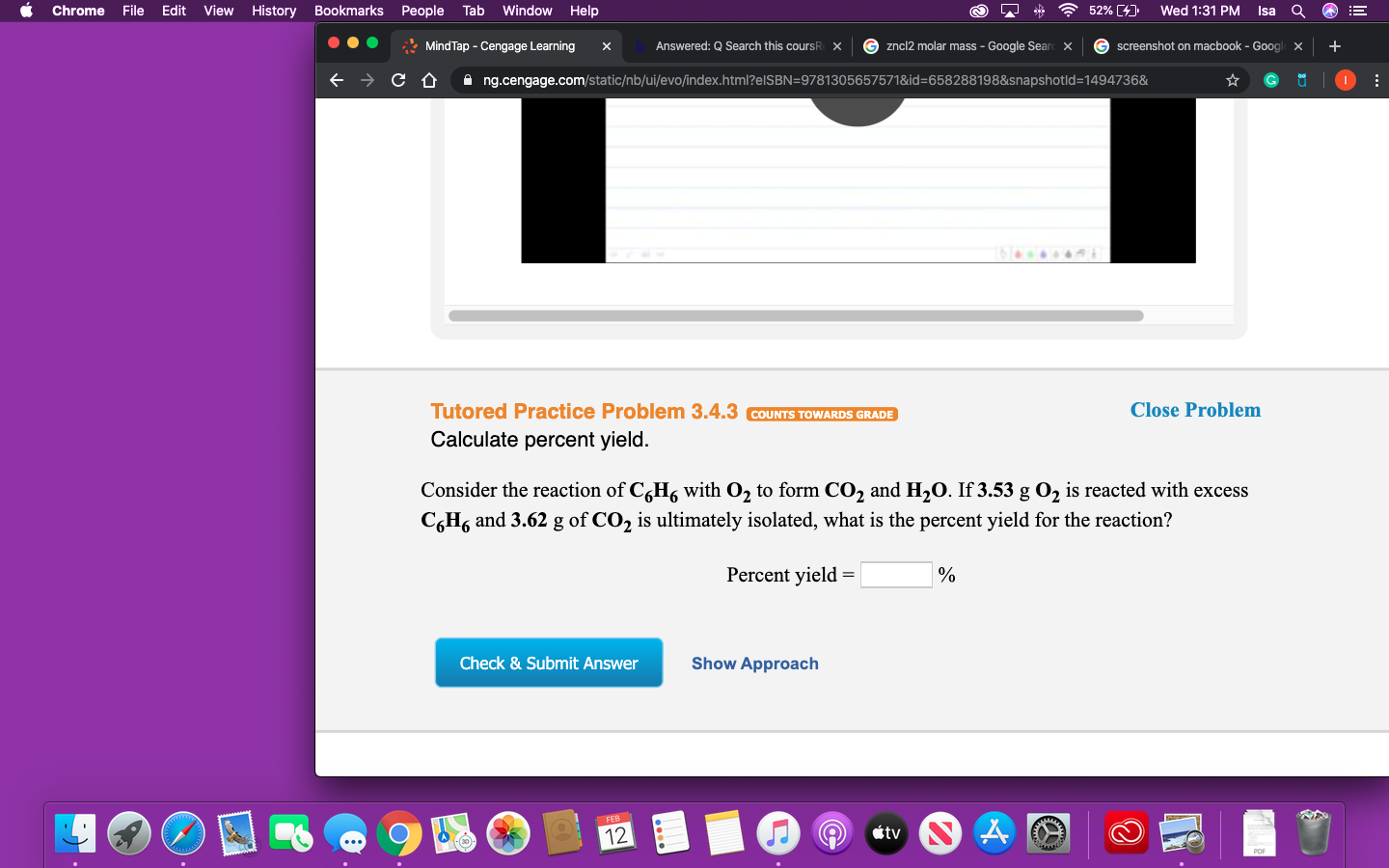
Transcribed Image Text:52% (4)
File
Edit
View
History Bookmarks People
Tab
Window Help
Wed 1:31 PM
Chrome
Isa
* Mind Tap - Cengage Learning
Answered: Q Search this coursR
G zncl2 molar mass - Google Sear
G screenshot on macbook - Google x
X
A ng.cengage.com/static/nb/ui/evo/index.html?elSBN=9781305657571&id=658288198&snapshotld=1494736&
Tutored Practice Problem 3.4.3 COUNTS TOWARDS GRADE
Close Problem
Calculate percent yield.
Consider the reaction of C,H, with O, to form CO2 and H2O. If 3.53 g O2 is reacted with excess
C,H6 and 3.62 g of CO2 is ultimately isolated, what is the percent yield for the reaction?
Percent yield =
Check & Submit Answer
Show Approach
FEB1
étv
12
POF
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

