Q: 5- In each part, choose the structure that best adapts to the infrared spectrum
A: g) In this case, the structure should be Reason: simply base value for isolated aldehyde peaks in I...
Q: Calculate the pH of a 1.30 M CH3NH3Cl solution. Kb for methylamine, CH3NH2, is 4.4 × 10-4. Answer is...
A: The value of Kb for the given weak base, CH3NH3Cl is = 4.4x10-4 The concentration of the base, CH3NH...
Q: QUESTION 3 Isopropyl alcohol, C3H7OH, is a nonelectrolyte. A 154.00 g sample of isopropyl alcohol is...
A: Given, Mass of isopropyl alcohol = 154.00 g Mass of water solvent = 250.00 g = 0.250 Kg Note: Van't...
Q: Question 5: Which manganese species are unstable with respect to disproportionation in aqueous solut...
A:
Q: How many liters are needed to make a 4.8M solution of KBr if 4 mole of KBr are available for use?
A:
Q: `NH2 What is the name of the structure shown?
A:
Q: Attempt 3 Some of the formulas could be either molecular or empirical formulas; however, some can on...
A: A molecular formula can be defined as the chemical formula that represents the total number of atoms...
Q: (CH3)2-CH-CH(CH3)-CH2-CH(C6H5)-CH2-CH2-CH3 O 2,2-Dimethyl-octyl-benzene O 2,3-Dimethyl-5-phenyl-octa...
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: Required is 500 cc of 4/5 (v/v) strength solution. The usual solution is 3% (v/v) hydrogen peroxide ...
A:
Q: 15. Americium - 242 is a beta emitter. Write an equation for this nuclear reaction and identify the ...
A:
Q: Draw an achiral diastereomer of the molecule shown below CH3 Но -- OH H- OH CH3
A: An achiral compound is the compound that must have an superimposable mirror image. Again, If two co...
Q: Provide a detailed narrative description of the mechanism of carbonic anhydrase. your narratives des...
A:
Q: Consider the following reaction at 25°C with the ΔG°' = +1800 J/mol for the forward reaction. The mo...
A:
Q: I dentify the functional groups m the following structures, If a funcional gmp is frund more tham nc...
A: We have to tell the functional group present in the above compound which are L-Dopa and Sevin.
Q: The following reaction is monitored as a fraction of time: AB=A+B. A plot of 1/[AB] versus time yiel...
A:
Q: Consider this reaction: 2H,PO, (aq) - P,05 (aq)+3H,0 (aq) At a certain temperature it obeys this rat...
A:
Q: The ocean is approximately a 3.00% solution of NaCl (fw%3= Calculate the molarity of NaCl in the oce...
A:
Q: 1.Write the balanced, net ionic equation for the reaction occurring when (a) Cl is added to a soluti...
A:
Q: Current Attempt in Progress In the reaction, KHS(aq) + HCI(aq) → KCI(aq) + H2S(g), which ions are th...
A: The given reaction is, KHS(aq) + HCl(aq) ---> KCl(aq) + H2S(g) The ionic equation for the given...
Q: a The H3C-C | at CCH2 The number of unshared pairs at atom a is The number of unshared pairs at atom...
A:
Q: 14. In the equation 22890Th 22891Pa + X which particle is represented by X? Accessibility. Good to g...
A:
Q: A certain solution consists of isopropyl alcohol diluted in water. If a 500 mL bottle contains 75 mL...
A: The volume of solute in milliliter present in 100 mL solution is known as the volume percent concent...
Q: Consider the same reaction carried out two more times, both at the same condition(including concentr...
A:
Q: Sketch a plot of the fraction of species, f, vs pH for Tyr. Show your calculations for several poin...
A: Answer -
Q: Objectives: a) Define Aufbau principle, Pauli Exclusion Principle, and Hund's Rule. b) List and desc...
A: Aufbau principal helps us to state how electrons are arranged in orbitals according to their energy....
Q: earn the term “oxime” and then look up a reaction called the “Beckmann rearrangement.” Why is this r...
A:
Q: At a certain temperature the rate of this reaction is second order in NH,OH with a rate constant of ...
A:
Q: ulometry is based on measu number of electrons that ticipate in a chemical react ventional titration...
A: We know that: Coulometry is based on an exhaustive electrolysis of the analyte i.e the analyte is co...
Q: Sodium only has one naturally occuring isotope, 23Na, with a relative atomic mass of 22.9898 u. A sy...
A: Given, 22Na has a relative atomic mass of 21.9944 u. 23Na, with a relative atomic mass of 22.9898 u....
Q: If I have a 7M HCl solution, how many mL are needed from the stock solution to prepare 54 mL of a 2M...
A:
Q: 1-2) The Atomic Hotel The Atomic Hotel is a special hotel designed for electrons. The hotel has a st...
A: We need to determine the electrons which do not follow Aufbau principle and Pauli Exclusion principl...
Q: The following reaction is monitored as a function of time: AB=A+B. A plot of 1/[AB] versus time yiel...
A:
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the fo...
A:
Q: A certain solution is 2.5% (w/v) NaCl (salt) in water. Calculate the number of grams of sodium chlor...
A:
Q: Which of the following C5H8 compounds best fits the spectrum. The integration values are shown above...
A: By correlating chemical shift values and splitting of proton NMR signal with the individual proton a...
Q: The rate at which a certain drug is eliminated by the body follows first-order kinetics, with a half...
A:
Q: 16. Bismuth - 210 is an alpha emitter. Write an equation for this nuclear reaction and identify the ...
A: The given species is Bismuth-210.
Q: 4. Complete and balance the following half-reactions. In each case, indicate whether the half-reacti...
A:
Q: What is the molar mass for calcium chloride dihydrate? What is the molar mass of calcium oxalate mon...
A: We have to calculate the molar mass of the given compounds. The given compounds are Calcium chlorid...
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature k ...
A: Here we are given.,
Q: Electrophilic addition of HI
A:
Q: What is the purpose of refluxing the reaction mixture for 45 minutes? Why not simply boil the mixtur...
A: A question based on analytical process that is to be accomplished.
Q: ing statements is true of the detection limit of an instrument?
A: The first question is based on antiquity, so instead of solving it, solve the second question with t...
Q: Give the nuclear symbol (isotope symbol) for the isotope of bromine, Br, that contains 46 neutrons p...
A: an atom consist of three subatomic particle proton, neutron, electron here we are required to find t...
Q: For each system listed in the first column of the table below, decide (if possible) whether the chan...
A:
Q: Give two examples of common radioisotopes used in medicine. Be sure to write the full name of the is...
A: In medical world radioisotopes used in various ways.Radioisotopes can occurred as naturally or artif...
Q: 8. Identify the inhibitor from the following list for the selective target of the enzyme having C in...
A: o Acrylamide groups act as Michael acceptors and thus react with nucleophiles like thiol (SH) groups...
Q: Degradation of an airport's aging fuel tanks is suspected, resulting in both rust and water contamin...
A: A question based on properties of liquids that is to be accomplished.
Q: Choose the electron configuration that is most appropriate for nitrogen monochloride (NCl). Choose...
A: The atomic number of nitrogen atom is 7. It has 5 electrons in its outermost shell. The atomic numbe...
Q: Answer the below questions and show steps: a. You have a 50X stock of buffer and you need 150 µl of...
A:
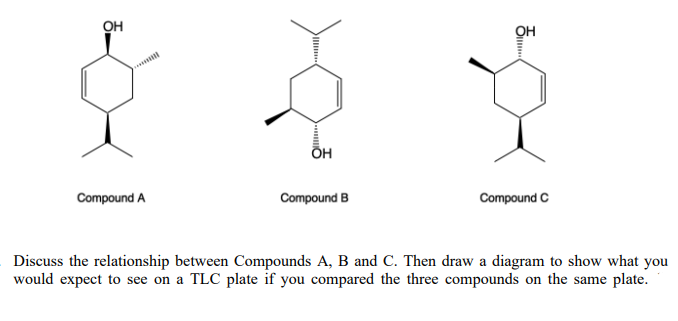
Step by step
Solved in 2 steps with 1 images

- a. Calculate the Rf value for each spot on the TLC plates below. Make sure you label which Rf belongs to which compound. Which compound corresponds to the unknown? b. Which compound corresponds to the unknown? Explain your answer.identify and interpret the peaks and their characteristics (C=O, 4H, doublet) product name: dulcin (4-ethoxyphenylurea) (C9H12N2O2) images include CNMR, HNMR, and IR spectroscopyN1. Which of the following molecule pair(s) could be potentially distinguished by TLC analysis?
- 1. Which compound will have the largest Rf value on a TLC plate using 10% ethyl acetate/hexane as a developing solvent: 3- decanone or 3- decanol, toluene or benzoic acid, cyclohexane or cyclooctanone? Explain why?Identify the Brassicasterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.Pretending that you got the following TLC result: A. Which is more polar? SA or C? B. Is the extraction that gave the TLC plate in the picture, successful or not? Explain 
- Whagt is the purpose of the following? Separate the active ingredients in an unknown over the counter analgesic using TLC (thin layer chromatography). Make a preliminary identification of each active ingredient in an unknown tablet by comparing its Rf value(s) with the Rf values of four different known active ingredients:I had having trouble understanding the TLC labs. Could someone help explain how to tell which compound has a higher Rf value? benzoic acid or trans-stilbene?Identify the Campesterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.
- Which of the compounds listed below would have the highest Rf value in a TLC experiment? A) naphthaleneB) acetylsalicylic acid C) ascorbic acidD) 1-naphtholCompound A had an Rf value of 0.35 in pentane and 0.45 in diethyl ether when generated on a TLC plate using pentane. Compound B exhibits Rf 0.42 in pentane and 0.60 in diethyl ether. What solvent should be used for the TLC separation of an A/B mixture? Explain.Draw the structure that fits each spectrum and assign each CHx group to appropriate peaks. (reference point at 0 ppm)



