Show that the third Bohr postulate, Eq. (2–5), is equivalent to an integer num- ber of de Broglie waves fitting within the circumference of a Bohr circular orbit.
Show that the third Bohr postulate, Eq. (2–5), is equivalent to an integer num- ber of de Broglie waves fitting within the circumference of a Bohr circular orbit.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter10: Introduction To Quantum Mechanics
Section: Chapter Questions
Problem 10.53E: Show that the normalization constants for the general form of the wavefunction =sin(nx/a) are the...
Related questions
Question
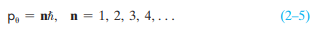
Transcribed Image Text:ni, n = 1, 2, 3, 4, ...
(2–5)

Transcribed Image Text:Show that the third Bohr postulate, Eq. (2–5), is equivalent to an integer num-
ber of de Broglie waves fitting within the circumference of a Bohr circular
orbit.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,