Q: Show how to convert 4-methoxybenzyl chloride to each amine.
A: The given reagent is 4-methoxybenzyl chloride. The product that is to be formed is represented as…
Q: 2. Complete the retrosynthetic analysis of compound B. Leave FGI blank if no functional group…
A: FGI is the functional group inter conversion where one functional group is interconverted to other…
Q: Propose a mechanism for the following reaction:
A: This is a reaction of aldehydes with amines, where amine nitrogen atom attacks the carbonyl carbon,…
Q: i. butanoic anhydride + sodium hydroxide j. butanoyl bromide + potassium ethoxide chs-CHz- CHz- ċ-Br…
A: When an acid or its derivative reacts with alcohol or its derivative, there is a formation of ester…
Q: Draw the products of the pentanoic acid + PBr3 + Br2, followed by hydrolysis
A: The reaction is as follows:
Q: Most Activating 2 Least Activating GOH3 OH Oph CH Pheno? anisure 13r |-naphthol Diphenyl etler…
A: This question is checking the basic concept of nature of Benzylic lone pairs. We have to tackle…
Q: Do a retrosynthetic analysis on the following compound, ending withavailable starting material.
A: In a retrosynthetic analysis, in order to prepare the target compound, the precursor is identified…
Q: Multistep synthesis from phenol to anethole?
A: Conversion of phenol to anethole (1-Methoxy-4-[(1E)-prop-1-en-1-yl]benzene) is a multistep synthesis…
Q: Use the malonic ester synthesis to prepare each carboxylic acid.
A: Melonic ester synthesis method is used to synthesise carboxylic acids with desired carbon chain.
Q: Complete the reaction and draw: Acetophenone reacts with phenylmagnesiumbromide in presence of H2O…
A: Given reaction condition: Acetophenone reacts with phenylmagnesiumbromide in presence of H2O and…
Q: Show the product of treating this anhydride with reagent
A: Given: Treatment of given anhydride with methanol
Q: Draw the product formed when pentanoic anhydride [(CH;CH,CH,CH,cO),0] is treated with each reagent.…
A: Given : Draw the product formed when pentanoic anhydride is treated with each reagent. a. SOCl2 b.…
Q: Propose a mechanism for the base-catalyzed aldol condensation of acetone (Figure 22-2).S
A:
Q: Why could you not synthesize phenacetin through a Williamson ether synthesis using the following…
A: Williamson ether synthesis- Reaction between alkoxide and alkyl halide to form ether. Reaction goes…
Q: Show how to synthesize the following compound using either the malonic ester synthesis or the…
A: 4-Phenyl-2-butanone: Its common name is benzylacetone. It is flowery in smell. It is prepared by…
Q: *Pu OK it 90 °C
A: An aldol condensation is a condensation reaction in which an enolate ion reacts with a carbonyl…
Q: Propose a synthetic route to prepare the same ketone in four steps starting from…
A: Organic reaction mechanisms
Q: H HO acetaminofen
A: To provide: The synthesis of benzene from acetaminophen.
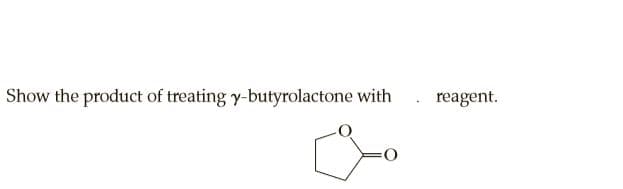
Q: Show the product of treating y-butyrolactone with reagent.
A: The given ester reacts with hydroxide ion and forms carboxylic acid which again reacts with the base…
Q: a OH CH2CNHCH3 CH3 C d OH CHCHNHCH3 CH3 Ephedrine can be synthesized via reductive amination plus…
A: Here we have to predict necessary reactants and reagents that will give the intermediate and final…
Q: Use the malonic ester synthesis to prepare each carboxylic acid. .COOH a. CH;CH,CH2CH¿CH¿CH2COOH b.…
A: Malonic ester is an dicaebonyl compound in which -CH2 group is sandwitched between -CO2Et group.…
Q: show and explain the mechanism with 3-nonenoic acid to y-nonanoic lactone?
A: The reaction of the conversion of 3-nonenoic acid to nonanoic lactone is the conversion of a…
Q: 1. 2 CH,Li, ether 2. H,O, HCI
A:
Q: Show how to convert each starting material into benzylamine in good yield
A: Given starting material,
Q: ČOEt 1. EtO-Na+ OEt 3. NAOH, H,O, heat EtO 4. HCI, heat 2-Carbethoxy- 4-Butanolactone…
A:
Q: How might you convert cyclopentyl carboxylic acid to N-isopropyl cyclopentylcarboxylamide? Give two…
A: Answer - Add N,N′-dicyclohexylcarbodiimide(DCC) along with Isopropyl amine, which will carry out…
Q: He OH et worle HO
A: An acetal is a compound that has two ether-like -OR groups bonded to the same carbon atom of what…
Q: Show the products from the reaction of p-bromoaniline with following reagents: a)CH3Cl(excess) b)…
A: In p-bromoaniline due to the presence of NH2 group, the compound is basic in nature, thus it does…
Q: tautomerization OH
A: Lindlar catalyst always gives cis alkene
Q: Propose a one-step synthetic route to obtain 1-octyne from sodium acetylide.
A: Sodium acetylide can be used to synthesize alkynes using alkyl halide. They acts as strong bases and…
Q: Match the following reactions with the appropriate reagent, intermediate, or product. Match each…
A: A reagent is a compound that is added to a system to cause a chemical reaction, or test if one…
Q: Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some…
A: When the above given compound reacts with LiAlH4. it is reduced to alcohols.LiAlH4 provides 4…
Q: Show the product of treating the following g-lactam with reagent
A: The reagent, Lithium aluminum hydride is a reducing agent. It reduces amide functional group to…
Q: Show the product of treating y-butyrolactone with reagent.
A: Lactones are esters formed by various organic acids which are cyclic in nature. Gamma lactones are…
Q: Show how to synthesize the following compound using either the malonic ester synthesis or the…
A: Acetoacetic ester synthesis is a chemical reaction where ethyl acetoacetate is alkylated at the…
Q: Show the reaction mechanism of 2-butanone with the appropriate reagents to obtain an enamine.
A:
Q: la. Show the reaction needed to prepare propyl phenyl ketone from CH,CH,CN and a Grignard reagent.…
A: Grignard reaction: It is an organometallic chemical reaction in which alkyl or aryl magnesium…
Q: Show how to synthesize the following compound using either the malonic ester synthesis or the…
A: The malonic ester synthesis of 2-Propyl-1,3-propanediol undergoes a nuclophilic attack using strong…
Q: Sodium borohydride (NABH4) is a very selective reagent. Which functional groups can sodium…
A: Sodium borohydride (NaBH4 ) is a week reducing agent.
Q: Show the product of treating this anhydride with reagent
A:
Q: Propose a mechanism for the aldol reaction below 2 H3C H OH-, H₂O H3C H
A:
Q: i. 8-valerolactone j. 2-chloroethanoyl chloride k. formaldehyde 1. a-oxo valeraldehyde…
A:
Q: 1. LIAIH, 2. H2O
A: LiAlH4 is a reducing agent which converts C=O group to CH2 group.
Q: Propose a correct retrosynthetic analysis of (E)-1,3-di(furan-2-yl)prop-2-en-1-one following an…
A: Aldol condensation : Alpha H containing carbonyl compounds in presence of dil. base gives beta…
Q: An amide with an NH2 group can be dehydrated to a nitrile with thionyl chloride (SOCl2). Propose a…
A: Thionyl chloride when reacts with 1° amide, a nitrile can be produced. A four step mechanism can be…
Q: The naturally occurring lactone Sedanenolide is the major contributor to the flavor of celery. Amend…
A:
Q: Please draw out the 2 reaction schemes: Ethyl diacetoacetate + hydrazine = pyrazole Ethyl…
A:
Q: Give the plan for the next retrosynthesis. Other compounds may be used. All reagents must be…
A: To get the product from the starting compound, need the following reactions 1) Dehydration : Removal…
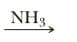
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Given this retrosynthetic analysis, propose a synthesis for fexofenadine from the four named starting materialsGiven this retrosynthetic analysis, propose a synthesis of proparacaine from 4-hydroxybenzoic acidPropose a mechanism for the reaction of benzoic acid with acetyl chloride to giveacetic benzoic anhydride

