Reactions of Ethers
Ethers (R-O-R’) are compounds formed by replacing hydrogen atoms of an alcohol (R-OH compound) or a phenol (C6H5OH) by an aryl/ acyl group (functional group after removing single hydrogen from an aromatic ring). In this section, reaction, preparation and behavior of ethers are discussed in the context of organic chemistry.
Epoxides
Epoxides are a special class of cyclic ethers which are an important functional group in organic chemistry and generate reactive centers due to their unusual high reactivity. Due to their high reactivity, epoxides are considered to be toxic and mutagenic.
Williamson Ether Synthesis
An organic reaction in which an organohalide and a deprotonated alcohol forms ether is known as Williamson ether synthesis. Alexander Williamson developed the Williamson ether synthesis in 1850. The formation of ether in this synthesis is an SN2 reaction.
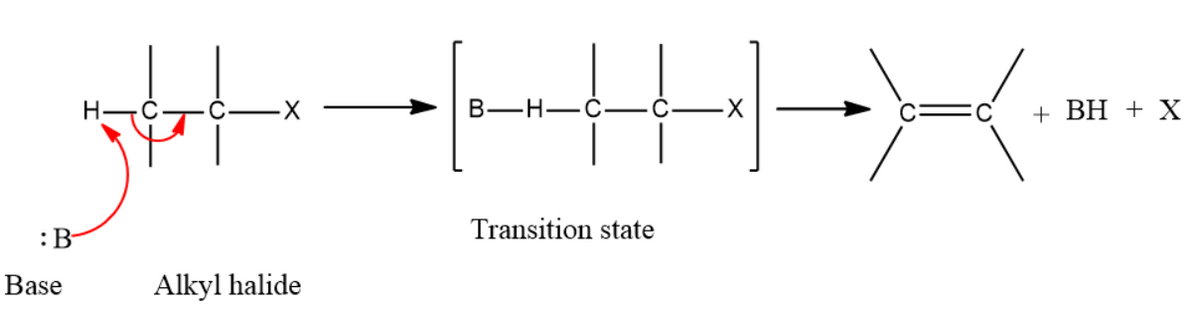
Show the relationship between E2 Reactions and
Interpretation: The relationship between E2 reactions and alkyne synthesis is to be shown.
E2 reactions:
In an E2 mechanism which refers to bimolecular elimination is basically a one-step mechanism.
Here, the carbon-hydrogen and carbon-halogen bonds mostly break off to form a new double bond.
However, in the E2 mechanism, a base is part of the rate-determining step and it has a huge influence on the mechanism.
General mechanism of E2 reaction is as shown below,
Alkyne synthesis:
In general, chlorine or bromine is used with an inert halogenated solvent like chloromethane to create a vicinal dihalide from from an alkene. The vicinal dihalide formed is the reactant needed to produce the alkyne using double elimination.
Mechanism:
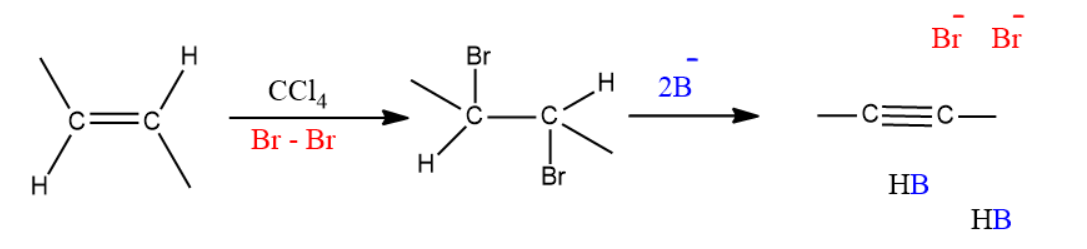
Step by step
Solved in 3 steps with 2 images


