Shown below is a common lipid. To which of the following lipid groups does this molecule belong? CH₂ CH₂ ICH Triacylglycerol Ceramide Cholesterol Ganglioside Glycerophospholipid Monounsaturated fatty acid Polyunsaturated fatty acid Saturated fatty acid
Shown below is a common lipid. To which of the following lipid groups does this molecule belong? CH₂ CH₂ ICH Triacylglycerol Ceramide Cholesterol Ganglioside Glycerophospholipid Monounsaturated fatty acid Polyunsaturated fatty acid Saturated fatty acid
Chapter4: Lipids
Section: Chapter Questions
Problem 3SC
Related questions
Question
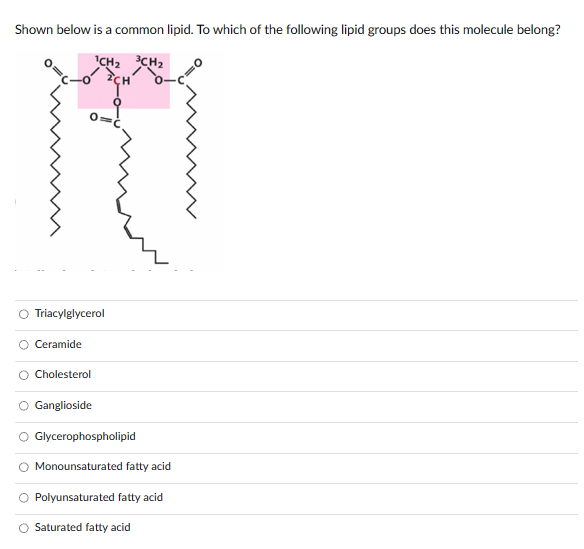
Transcribed Image Text:Shown below is a common lipid. To which of the following lipid groups does this molecule belong?
Triacylglycerol
Ceramide
Cholesterol
CH₂ CH₂
Ganglioside
Glycerophospholipid
Monounsaturated fatty acid
Polyunsaturated fatty acid
Saturated fatty acid
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning