Sickle cell anemia is caused by too much consumption of disaccharides and monosaccharides a change in a single amino acid in the peptide sequence reaction of lipase to the blood cell, causing disruption in the blood flow a virus similar to coronavirus that have a protein similar to Sars-Cov2 Since cholesterol is a lipid, that would make it insoluble in blood. There is a correlation between high blood pressure and this molecule since O it clogs arteries and at high levels form plaque blood pressure becomes low from the IMF between these substances lipids are mostly hydrophilic, causing denaturation in the protein in blood the "good" and "bad" cholesterol are interchangeable and has a high pH
Sickle cell anemia is caused by too much consumption of disaccharides and monosaccharides a change in a single amino acid in the peptide sequence reaction of lipase to the blood cell, causing disruption in the blood flow a virus similar to coronavirus that have a protein similar to Sars-Cov2 Since cholesterol is a lipid, that would make it insoluble in blood. There is a correlation between high blood pressure and this molecule since O it clogs arteries and at high levels form plaque blood pressure becomes low from the IMF between these substances lipids are mostly hydrophilic, causing denaturation in the protein in blood the "good" and "bad" cholesterol are interchangeable and has a high pH
Chapter5: Ground Rules Of Metabolism
Section: Chapter Questions
Problem 8SA
Related questions
Question
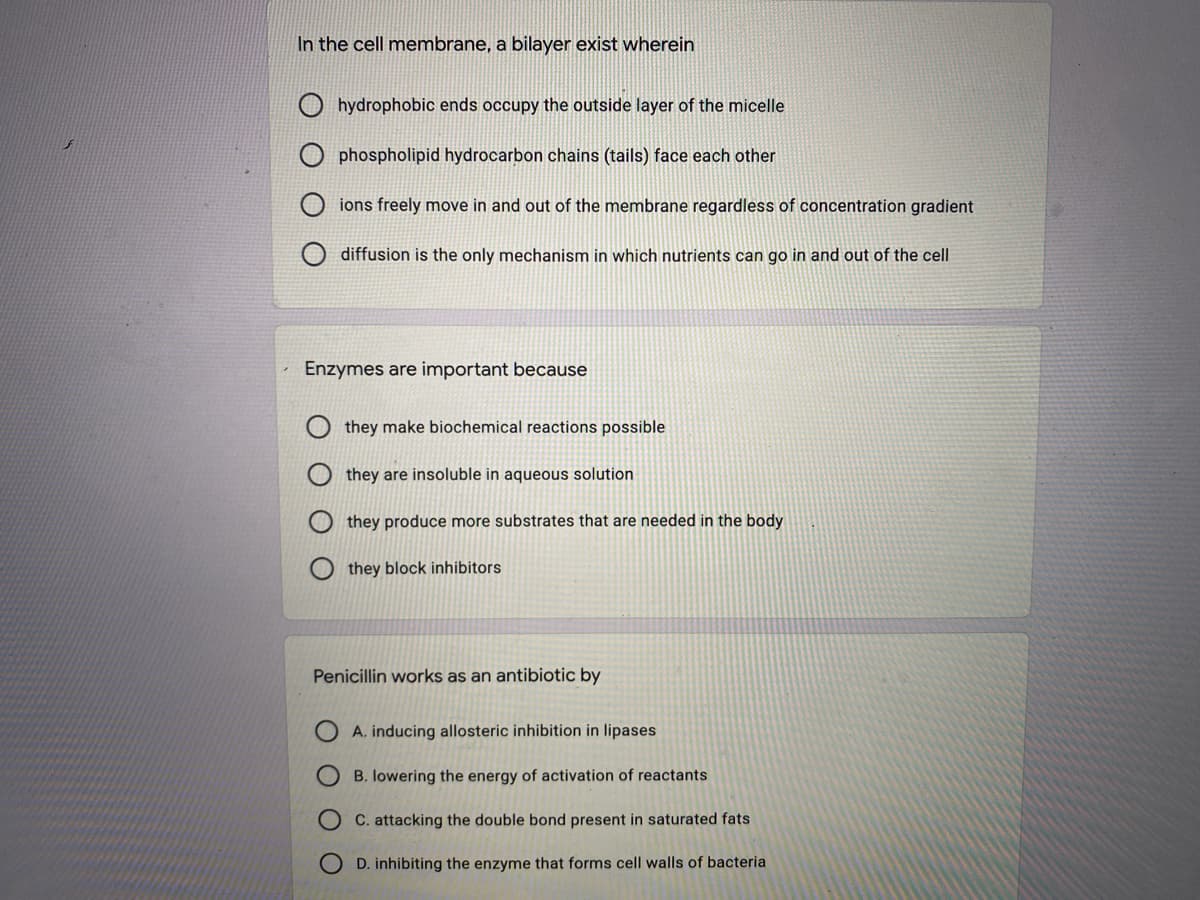
Transcribed Image Text:In the cell membrane, a bilayer exist wherein
hydrophobic ends occupy the outside layer of the micelle
O phospholipid hydrocarbon chains (tails) face each other
ions freely move in and out of the membrane regardless of concentration gradient
diffusion is the only mechanism in which nutrients can go in and out of the cell
Enzymes are important because
they make biochemical reactions possible
they are insoluble in aqueous solution
they produce more substrates that are needed in the body
they block inhibitors
Penicillin works as an antibiotic by
A. inducing allosteric inhibition in lipases
B. lowering the energy of activation of reactants
C. attacking the double bond present in saturated fats
D. inhibiting the enzyme that forms cell walls of bacteria
O O
O O O
O O O
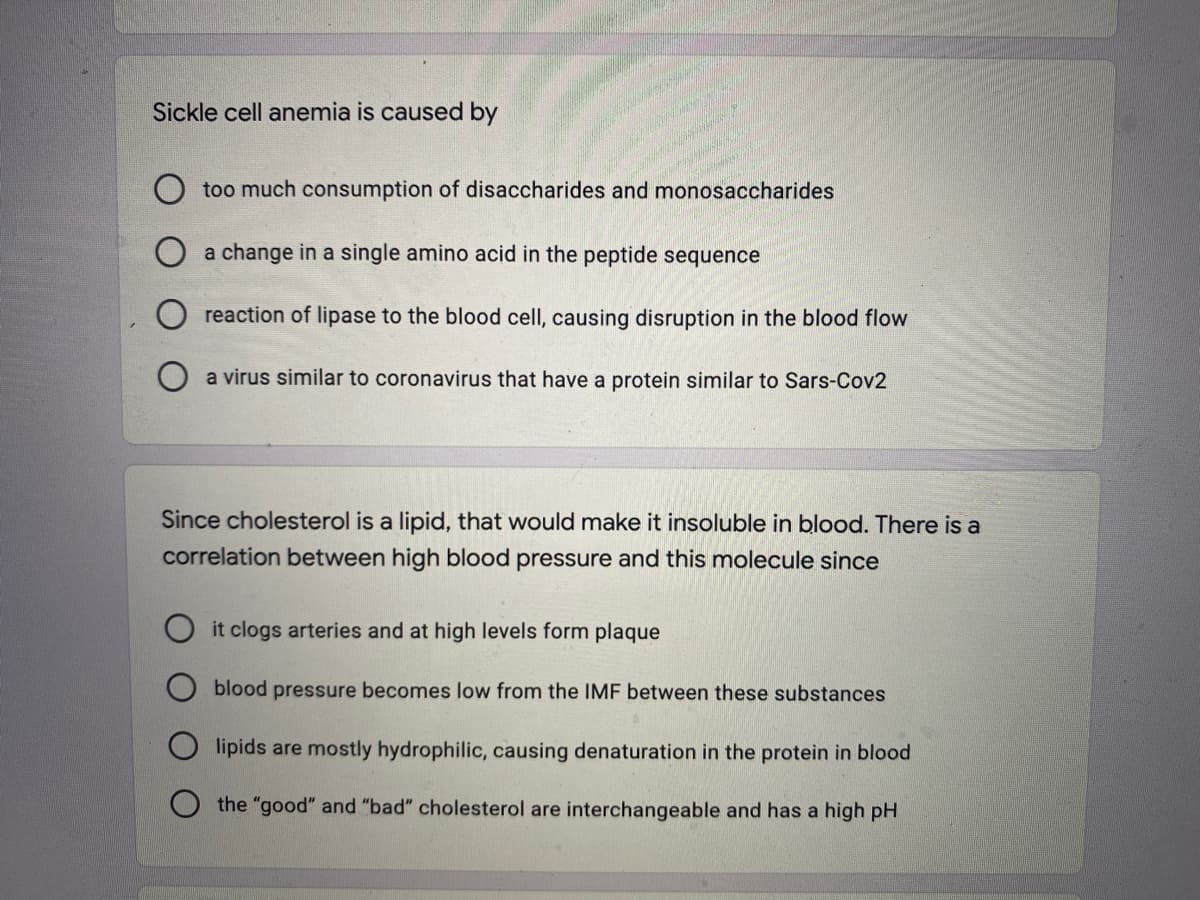
Transcribed Image Text:Sickle cell anemia is caused by
too much consumption of disaccharides and monosaccharides
a change in a single amino acid in the peptide sequence
reaction of lipase to the blood cell, causing disruption in the blood flow
a virus similar to coronavirus that have a protein similar to Sars-Cov2
Since cholesterol is a lipid, that would make it insoluble in blood. There is a
correlation between high blood pressure and this molecule since
it clogs arteries and at high levels form plaque
blood pressure becomes low from the IMF between these substances
lipids are mostly hydrophilic, causing denaturation in the protein in blood
the "good" and "bad" cholesterol are interchangeable and has a high pH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning