Siven the unbalanced chemical equation, C5H12(1) + O2(g) → CO(g) + H20(1), which one of the pllowing correctly expresses the mole ratio between CO and oxygen? O 5 mole CO/11 mole O2 O 1 mole CO/1 mole O2 O 10 mole CO/11 mole O2 O 10 mole CO/6.5 mole O2
Siven the unbalanced chemical equation, C5H12(1) + O2(g) → CO(g) + H20(1), which one of the pllowing correctly expresses the mole ratio between CO and oxygen? O 5 mole CO/11 mole O2 O 1 mole CO/1 mole O2 O 10 mole CO/11 mole O2 O 10 mole CO/6.5 mole O2
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter2: Chemical Compounds
Section: Chapter Questions
Problem 105QRT
Related questions
Question
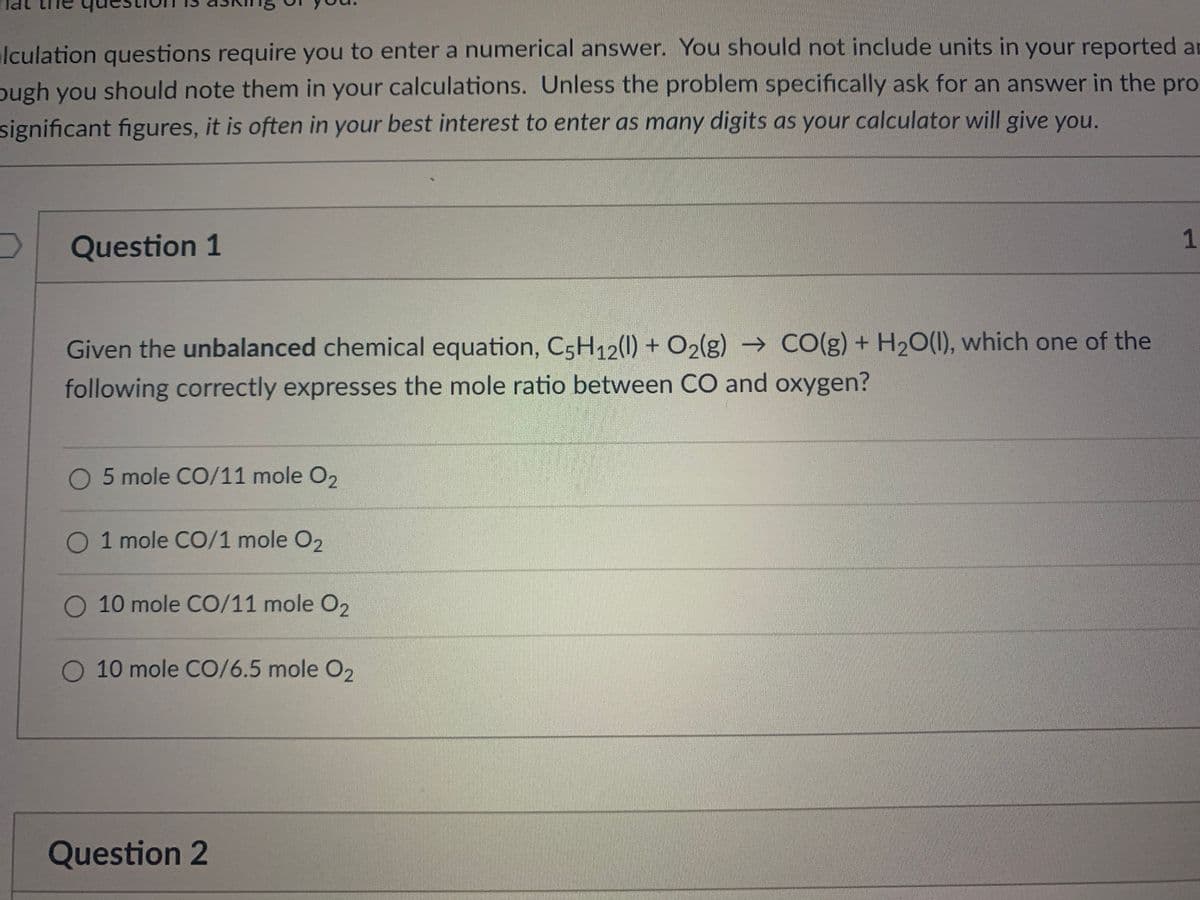
Transcribed Image Text:lculation questions require you to enter a numerical answer. You should not include units in your reported an
ough you should note them in your calculations. Unless the problem specifically ask for an answer in the pro
significant figures, it is often in your best interest to enter as many digits as your calculator will give you.
1
Question 1
Given the unbalanced chemical equation, C5H12(1) + O2(g) → CO(g) + H20(1), which one of the
following correctly expresses the mole ratio between CO and oxygen?
5 mole CO/11 mole O2
O 1 mole CO/1 mole O2
O 10 mole CO/11 mole O2
O 10 mole CO/6.5 mole O2
Question 2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning