Q: Write a chemical equation and a solubility product expression for the dissolution of dolomite in…
A: The chemical equation and a solubility product for the dissolution of dolomite in water it is to be…
Q: The aluminum in 0.200 g impure aluminum sulfate ( the sample) was precipitated with aqueous NH3and…
A: Since the weight of aluminum obtained form impure aluminum sulfate is 0.15 g. Molar mass of aluminum…
Q: What is the the equilibrium equation and the Ksp expression for CO2S3?
A:
Q: A 20.00 mL solution of 0.1200 M TI³+ is titrated with 0.2000 M Fe²+. TI³+ + 2e- TI* Eº = +1.280 V…
A: Given: Volume of Tl3+ = 20.00 mL Concentration of Tl3+ = 0.1200 M Concentration of Fe2+ = 0.200 M To…
Q: Explain why addition of NH3 or HNO3 to a saturated solution of Cu(OH)2 in contact with solid Cu(OH)2…
A:
Q: Using the equilibrium constant expression calculate the [H3O] and the ph of a buffer solution in…
A:
Q: Write a balanced, net ionic equation for the precipitation reaction of CaCl2 and Cs3PO4in an…
A:
Q: Calculate the solubility of Co(OH)3 (s), Ksp = 2.5 x 10-43, in a solution at pH= 4.0
A: The equilibrium is,
Q: The solubility of Ag3PO4 is measured and found to be 1.92×10-3 g/L. Use this information to…
A: Ksp is the product of solubility of ions of that compound.
Q: Calculate the solubility (in g/L) of Aluminum sulfide (Al2S3) In 0.25 M Al+3 solution. (standard…
A:
Q: (a) Explain why Mg(OH)2 precipitates when CO32- ion isadded to a solution containing Mg2+. (b) Will…
A: GIVEN: Mass of Na2CO3 = 4.0 g volume of solution= 1.00 L
Q: Why is NH4 more likely to give a precipitate with PO4
A: The formation of Ammonium phosphate takes place as follows: The NH4+ ions (conjugate acid of NH3)…
Q: Compounds with low solubility will NOT precipitate when we prepare them in the laboratory. OA. True…
A: The statement given is "Compounds with low solubility will not precipitate when we prepare them in…
Q: Find out in detail about the formation mechanisms and conditions of precipitates.
A: Answer - Precipitates - Precipitate: Is a solid formed by a change in a solution, often due to a…
Q: Calculate the solubility in grams per 100 ml H2O for La(IO3)3 (s), Ksp= 6.1*10^-12 BaF2 (s), Ksp=…
A:
Q: Calculate the solubility of Mg(OH)2 in a solution buffered to a pH of 12.0. Ksp= 1.8 x 10-11
A:
Q: Calculate the solubility of Ni(OH) 2 in a solution buffered at pH = 8.75 (K sp = 1.6 x 10 -16 ).
A:
Q: gives solubilities of the following compounds in grams per 100 mL of water. Because these compounds…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: 1. 0.4960 g sample of CaCO3 (mw= 100) is dissolved in an acidic solution. The calcium is…
A: "Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: How many milligrams of NaOH must be added to 100.0 mL of a 1.25 x 10-9 M CeCl3 solution in order to…
A: Recall the dissociation of CeCl3 CeCl3→Ce3+ + 3Cl-Find concentrations of ions obtained from…
Q: Calculate the solubility (g/L) of Pbl2 in 1.0 M Pb(NO3)2 solution. а. 1.4 x 10-8 b. 1.2 x 10-4 C.…
A:
Q: 5.31e-08 mol/L, what is the Ksp at this temperature? Ksp = (b) It is found that 9.14e-08 g of…
A:
Q: The solubility of Ag3PO4 is measured and found to be 2.00×10-3 g/L. Use this information to…
A:
Q: Analysis of an equilibrium solution of silver chromate indicates that the silver concentration is…
A: We have Concentration of Ag+ to be 1.3*10-4 M, we have to calculate the value of Ksp .
Q: 0.28g 0f MBr is dissolved in distilled water and made up to 250mL mark in a volumetric flask. 25 mL…
A: The titration of MBr by AgNO3 is a double-displacement reaction, which is given by,…
Q: Calculate the solubility (in moles per liter) of Fe(OH)3 (Ksp = 4 x 10-38) in each of the following.…
A: Answer:- This question is answered by using the simple concept of calculation of molar solubility in…
Q: What is the minimum pH at which Cr(OH)3 will precipitate from a solution that is 0.086 M in Cr3+…
A: The Ksp for salt is be expressed as :
Q: Based from the given problem, What is the molecular weight of potassium biphthalate (C8H5KO4)? *…
A: Standardization:- It is a method used to determine the exact concentration of a solution. Some…
Q: The solubility of La(IO3)3 in a 0.71 M KIO3 solution is 1.0 ×10-7 mol/L. Calculate Ksp for La(IO3)3.
A: Ksp = S2 For two ions Ksp = 4S3 for three ions KIO3 = K(+) + IO 3(-) La (IO3)3 -----> La 3+ +…
Q: The solubility of COCO3 is measured and found to be 1.03x10-4 g/L. Use this information to calculate…
A:
Q: The solubility of Ag3PO4 is measured and found to be 1.99×10-3 g/L. Use this information to…
A:
Q: 1. When Pearl added (NH4)2CO3 solution to an unknown salt solution in basic conditions, the solution…
A: Here we have predict which cation will not precipitate as carbonate when ammonium carbonate is added…
Q: A: Calculate the solubility product constant for PbCl2, if 80.0 mL of a saturated solution of PbCl…
A: The solubility of a compound defines the dissolution of the compound in a given solution. The…
Q: Discuss the solubility product constant values of silver halides in accordance with HSAB theory.
A: Q-Discuss the solubility product constant values of silver halides in accordance with HSAB theory.
Q: Explain why the addition of NH3 or HNO3 to a saturated solution of Ag2CO3 in contact with solid…
A: The substance which accepts an electron pair from the medium is called Lewis acid whereas the…
Q: Calculate the solubility of Co(OH)3, Ksp=2.5*10^-43, in a solution at PH=4.0
A: The solubility reaction is given by Co(OH)3 (s) ------> Co3+ (aq) + 3 OH- (aq) since pH = 4…
Q: The equilibrium concentrations in a solution of AC5 were found to be [A] = 0.100 M, [C] = 0.0240 M,…
A: Complex formation reaction: A+5C↔AC5 …Kf Solubility equilibrium reaction: AB(s)↔A+B …Ksp
Q: The solubility of Ca(OH)2 is 1.3 x 10-5 g/L a) Write a balanced equation for the solubility…
A: a) Calcium hydroxide dissociates into calcium ion and hydroxide anion as follows: Ca(OH)2 ⇔ Ca2+ +…
Q: Ammonia and potassium iodide solutions are added to an aqueous solution of chromium(III) nitrate. A…
A: Given , CrO3 formed = 0.0203 g sample of compound = 0.105 g
Q: If the molar solubility of PbCrO4 at 25 oC is 5.48e-07 mol/L, what is the Ksp at this temperature?…
A: Given, a). If the molar solubility of PbCrO4 at 25 oC is 5.48e-07 mol/L, what is the Ksp at this…
Q: Chromium(III) hydroxide, Cr(OH)3, is highly water insoluble. Its molar mass is 103.02 g/mol. a.)…
A: As you have posted multiple parts and have not mentioned which part you want to be solved. Hence we…
Q: The solubility of Ag3PO4 is measured and found to be 2.00×10-3 g/L. Use this information to…
A: Given Solubility of Ag3PO4 = 2 × 10-3 gram/Liter Solubility Product ( Ksp ) = ?
Q: The solubility of AgBr is measured and found to be 1.04x10-4 g/L. Use this information to calculate…
A:
Q: The solubility of Cr(OH)3 is measured and found to be 1.31x10-6 g/L. Use this information to…
A:
Q: What is the solubility of Cd(OH)2 at a pH of 11.08? Ksp(Cd(OH)2) = 5.000e-15.
A: Equation involved :- Cd(OH)2= Cd2+ + 2OH-
Q: When solid Cd(OH), is added to a solution of 0.10 M Nal, some of it dissolves. Calculate the pH of…
A: To calculate the pH of the solution at equilibrium when solid Cd(OH)2 is added to a solution of 0.10…
Q: The amount of iron in a meteorite is determined by a redox titration using KMnO4 as the titrant. A…
A: The ratio of mass of solute to the mass of solution multiply by 100 is known as %w/w.
Q: A 5.70 g iron ore was dissolved in concentrated HCl. After the dissolution, the resulting solution…
A:
Step by step
Solved in 2 steps with 2 images

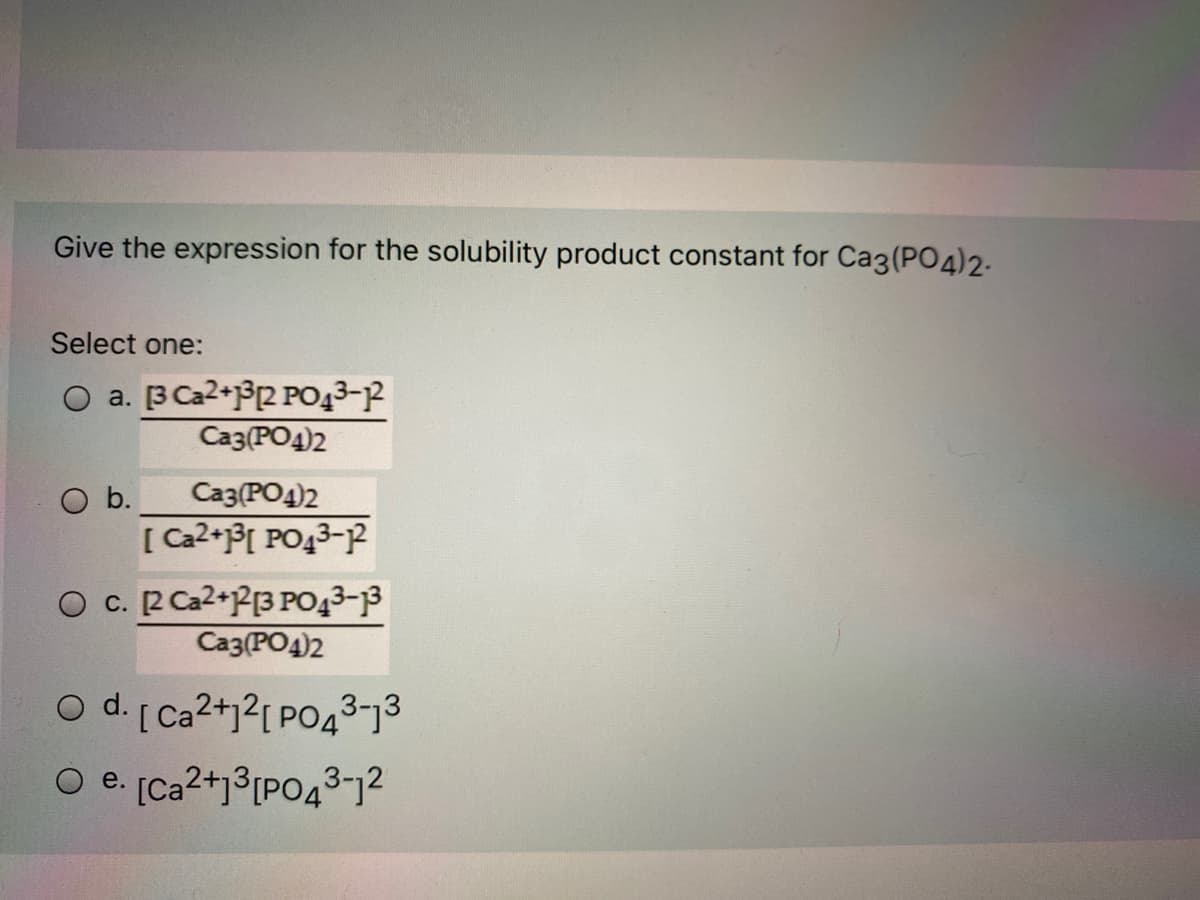
- Magnesium fluoride (MgF2, MW = 62.30 g/mol) is slightly soluble in water, with Ksp = 7.4 x 10^-11 at T= 25. C.a) Give the correctly balanced solubility reaction and expression for Ksp for MgF2.b) What mass of magnesium fluoride will dissolve in 1.000 L of pure water?1. a. When excess solid SrCrO4 is shaken with water at 25°C, it is found that 6 x 10^-3 mol dissolves per liter. Use this information to calculate the solubility product constant for SrCrO4. b. The Ksp of BaF2 is 1.8 x 10-7, what is its water solubility in moles per liter.16.The following is a solubility product expression for Ca3(PO4)2: Ksp= Group of answer choices A, [Ca32+][(PO43–)2] B, [Ca2+][2PO43–] C, [3Ca2+]3[2PO43–]2 D, [Ca3+]2[PO42–]3 E, [Ca2+]3[PO43–]2
- Calculate the solubility (in g/L) of silver chromate in water at 25°C if the Ksp for Ag3PO4 is 1.75 × 10-18. Ag3PO4(s) <==> 3Ag+ (aq) + PO43- (aq); Ksp = 27s4, s = _________ mol/L x MM of Ag3PO4 = _________ g/L.Calcium phosphate (Ca3(PO4)2; MM = 310.18 g/mol) has a Ksp of 2.07 × 10−33 at 25 ℃. Write the reaction equation for the dissolution of Ca3(PO4)2 solid Calculate the solubility in mg/L at 25 ℃ in pure water. Neglect other competing equilibria. Calculate the solubility in mg/L at 25 ℃ in the presence of 0.010 M CaCl2. Neglect other competing equilibria. Assume that 3s << 0.010 M. Is the assumption that s << 0.010 M valid? (Yes or No)At a particular temperature, the solubility of In₂(SO₄)₃ in water is 0.0070 M. You have found Ksp to be 1.8 × 10⁻⁹. If solid In₂(SO₄)₃ is added to a solution that already contains 0.200 M Na₂SO₄, what will the new solubility of the solid be? If solid In₂(SO₄)₃ is added to a solution that already contains 0.150 M ln(NO₃)₃, what will the new solubility of the solid be?
- a solution containing 6.25×10–3 M of S2– and 5.63×10–3 M of Cd2+ was prepared. CdS ⇌ Cd2+ + S2- Calculate the Qsp of the mixture (in 3 sig. figures) and In the given scenario, will a precipitate (Ksp = 3×10–28) form?The Ksp of ZnCO3 (MM: 125.38) is 1.20 xx 10^(-10). a) What is the molar solubility of ZnCO3? b) What is the solubility of ZnCO3 in g/L?The Ksp for CoCO3 = 1.0 × 10–10 and the Ksp for PbCO3 = 7.4 × 10–14. When Na2CO3 is added dropwise to a solution containing 0.010 M Co2+ and 0.010 M Pb2+, which ion will precipitate first? What must the [CO32–] be so that the first ion to precipitate comes out of solution, but the second ion does not?
- What is the Cl- concentration just as Ag2CrO4 begins to precipitate when 1.0 M AgNO3 is slowly added to a solution containing 0.015 M Cl- and 0.015 M I-? Ksp for Ag2CrO4 is 1.2 x 10-12.25). The solubility of BiI3 (dissociates to Bi3+ ions and I– ions) is 0.00778 g/L. What is Ksp for this compound? (a) Calculate the solubility of BiI3 in mol/L. (b) Show the reaction of dissociation of BiI3. (c) Show the Ksp expression for BiI3. (d) Calculate the Ksp for this compound?Consider the dissolution of solid Ca3(PO4)2 in an aqueous solution of 0.2 M Ca(NO3)2. Identify which ONE of the following statements is INCORRECT. a. Ca2+ is the common ion. b. The Ksp expression for calculating the solubility of Ca3(PO4)2 is Ksp = 108s5 c. The value of the Ksp for Ca3(PO4)2 remains unchanged d. After addition of Ca3(PO4)2 the Ca2+ concentration remains constant e. All nitrate anions remain in solution



