Solution B is to solution A. This is because solution B has a greater concentration of in it than does solution A. Solution C has no solutes dissolved in it, therefore it is tc Solutions A and B. As the relative concentration of solutes in two solutions increases, of necessit relative concentration of water in the same two solutions |Solution A has a lower co
Solution B is to solution A. This is because solution B has a greater concentration of in it than does solution A. Solution C has no solutes dissolved in it, therefore it is tc Solutions A and B. As the relative concentration of solutes in two solutions increases, of necessit relative concentration of water in the same two solutions |Solution A has a lower co
Biology: The Unity and Diversity of Life (MindTap Course List)
15th Edition
ISBN:9781337408332
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Chapter5: Ground Rules Of Metabolism
Section: Chapter Questions
Problem 11SQ: Solutes tend to diffuse from a region where they are _______ concentrated to an adjacent region...
Related questions
Question
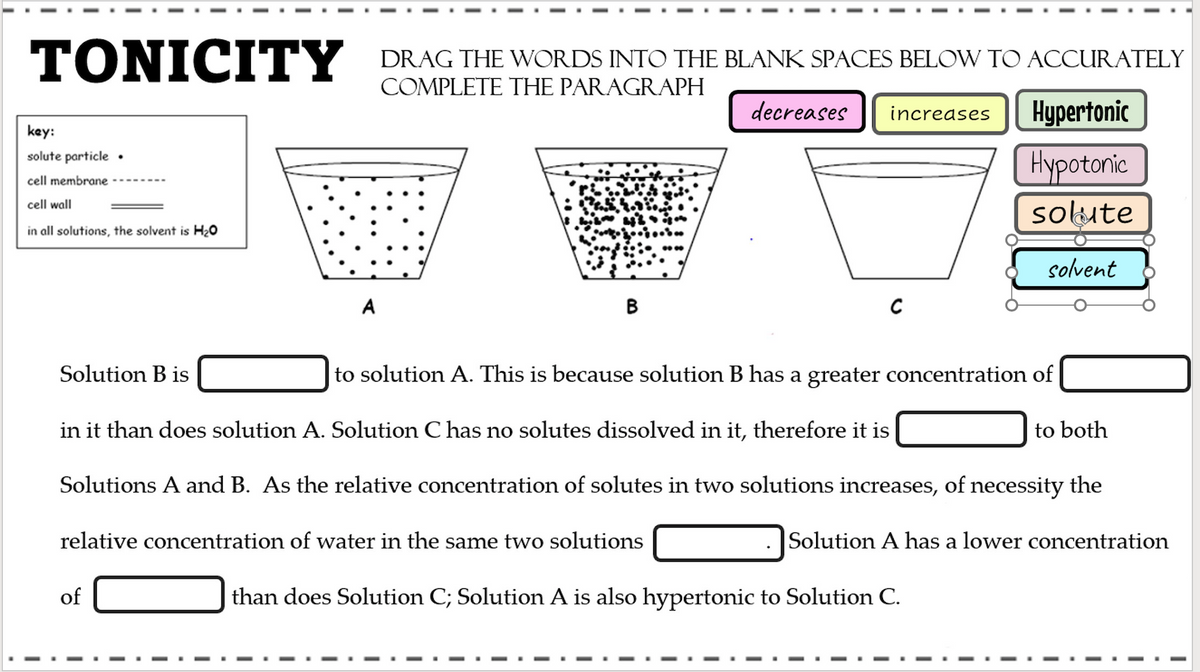
Transcribed Image Text:TONICITY DRAGTHE WORDS INTO THE BLANK SPACES BELOW TO ACCURATELY
COMPLETE THE PARAGRAPH
decreases
Hypertonic
increases
key:
Hypotonic
solute particle •
cell membrane
cell wall
solute
in all solutions, the solvent is H20
solvent
A
В
Solution B is
to solution A. This is because solution B has a greater concentration of
in it than does solution A. Solution C has no solutes dissolved in it, therefore it is
to both
Solutions A and B. As the relative concentration of solutes in two solutions increases, of necessity the
relative concentration of water in the same two solutions
Solution A has a lower concentration
of
than does Solution C; Solution A is also hypertonic to Sol
tion C.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning