gradient exists in a solution when the concentration of solutes in the solution is Da less than the amount of solvent. Ob. greater than the amount of solvent. Oc. the same everywhere. Od. not the same everywhere.
gradient exists in a solution when the concentration of solutes in the solution is Da less than the amount of solvent. Ob. greater than the amount of solvent. Oc. the same everywhere. Od. not the same everywhere.
Chapter12: Water Requirements For Aquaculture
Section: Chapter Questions
Problem 27SA
Related questions
Question
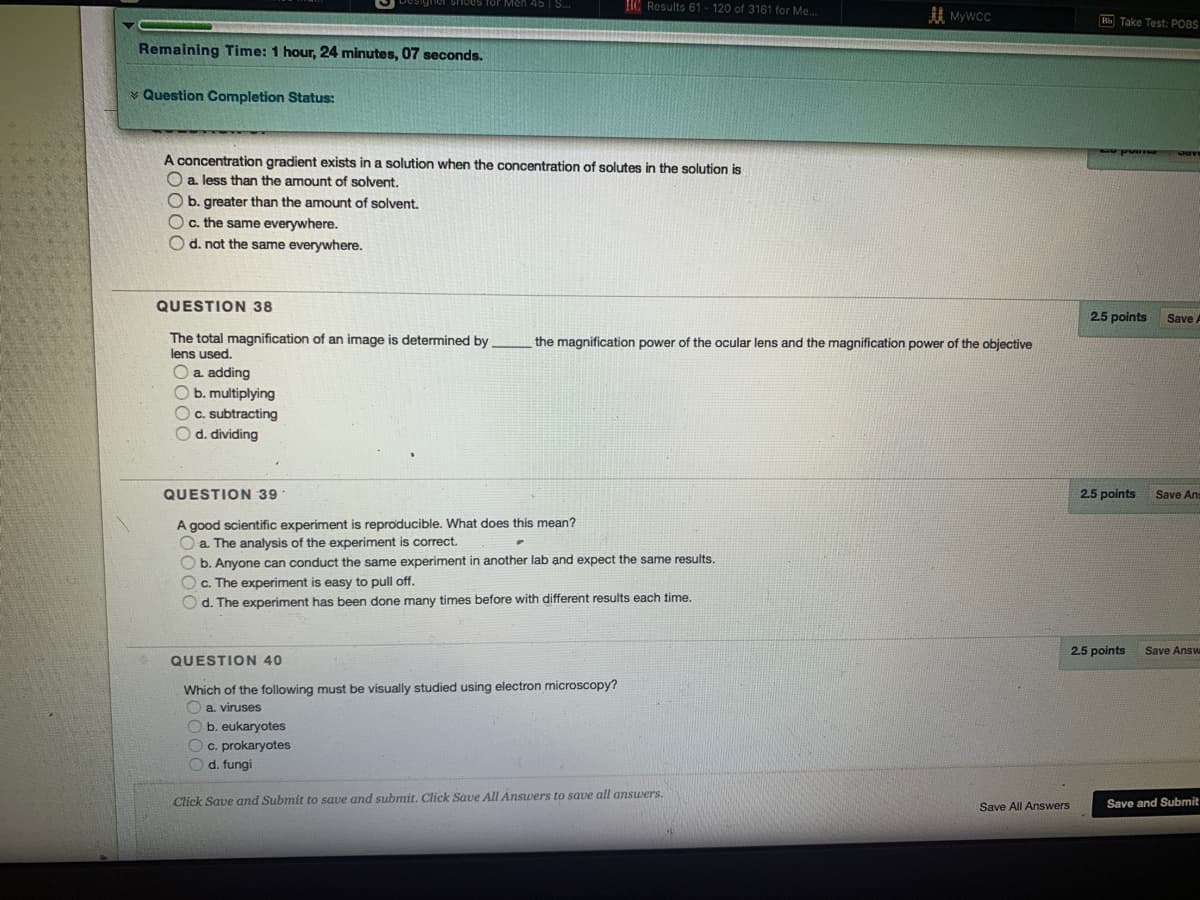
Transcribed Image Text:HC Results 61 - 120 of 3161 for Me.
从Mywcc
H Take Test: POBS
Remaining Time: 1 hour, 24 minutes, 07 seconds.
* Question Completion Status:
A concentration gradient exists in a solution when the concentration of solutes in the solution is
O a. less than the amount of solvent.
O b. greater than the amount of solvent.
Oc. the same everywhere.
O d. not the same everywhere.
QUESTION 38
2.5 points
Save.
The total magnification of an image is determined by
lens used.
O a adding
the magnification power of the ocular lens and the magnification power of the objective
O b. multiplying
O c. subtracting
O d. dividing
QUESTION 39
2.5 points
Save An
A good scientific experiment is reproducible. What does this mean?
O a The analysis of the experiment is correct.
O b. Anyone can conduct the same experiment in another lab and expect the same results.
O c. The experiment is easy to pull off.
O d. The experiment has been done many times before with different results each time.
2.5 points
Save Answ
QUESTION 40
Which of the following must be visually studied using electron microscopy?
a. viruses
O b. eukaryotes
O c. prokaryotes
O d. fungi
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
Save and Submit
Save All Answers
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you





