Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at nght). This makes it easy to isolate and measure the amount of gas produced. Suppose the O, gas evolved by a certain chemical reaction taking place at 30.0 Cs collected over water, using an apparatus something like that in the sketch, and the final volume of gas in the collection tube is measured to be 89.6 mL. Sketch of a gas-collection acparae Calculate the mass of O, that is in the collection tube. Round your answer to 2 significant digits. You can make any normal and reasonabie arpeon about the reaction conditions and the nature of the gases.
Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at nght). This makes it easy to isolate and measure the amount of gas produced. Suppose the O, gas evolved by a certain chemical reaction taking place at 30.0 Cs collected over water, using an apparatus something like that in the sketch, and the final volume of gas in the collection tube is measured to be 89.6 mL. Sketch of a gas-collection acparae Calculate the mass of O, that is in the collection tube. Round your answer to 2 significant digits. You can make any normal and reasonabie arpeon about the reaction conditions and the nature of the gases.
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter9: Gases
Section: Chapter Questions
Problem 70E: Before small batteries were available, carbide lamps were used for bicycle lights. Acetylene gas....
Related questions
Question
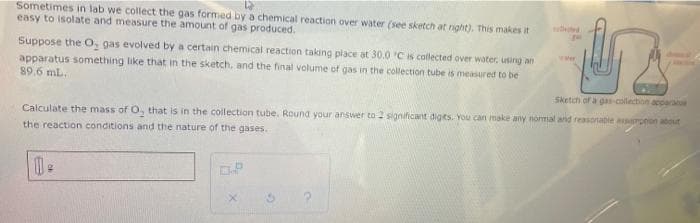
Transcribed Image Text:Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right). This makes it
easy to isolate and measure the amount of gas produced.
Suppose the O, gas evolved by a certain chemical reaction taking place at 30.0 Cis collected over water, using an
wer
apparatus something like that in the sketch, and the final volume of gas in the collection tube is measured to be
89.6 mL.
Sketch of a gas-collection acpara
Calculate the mass of O that is in the collection tube. Round your answer to 1 significant digits. You can imake any normal and reasonable a pnon about
the reaction conditions and the nature of the gases.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning