Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.7QAP
Related questions
Question
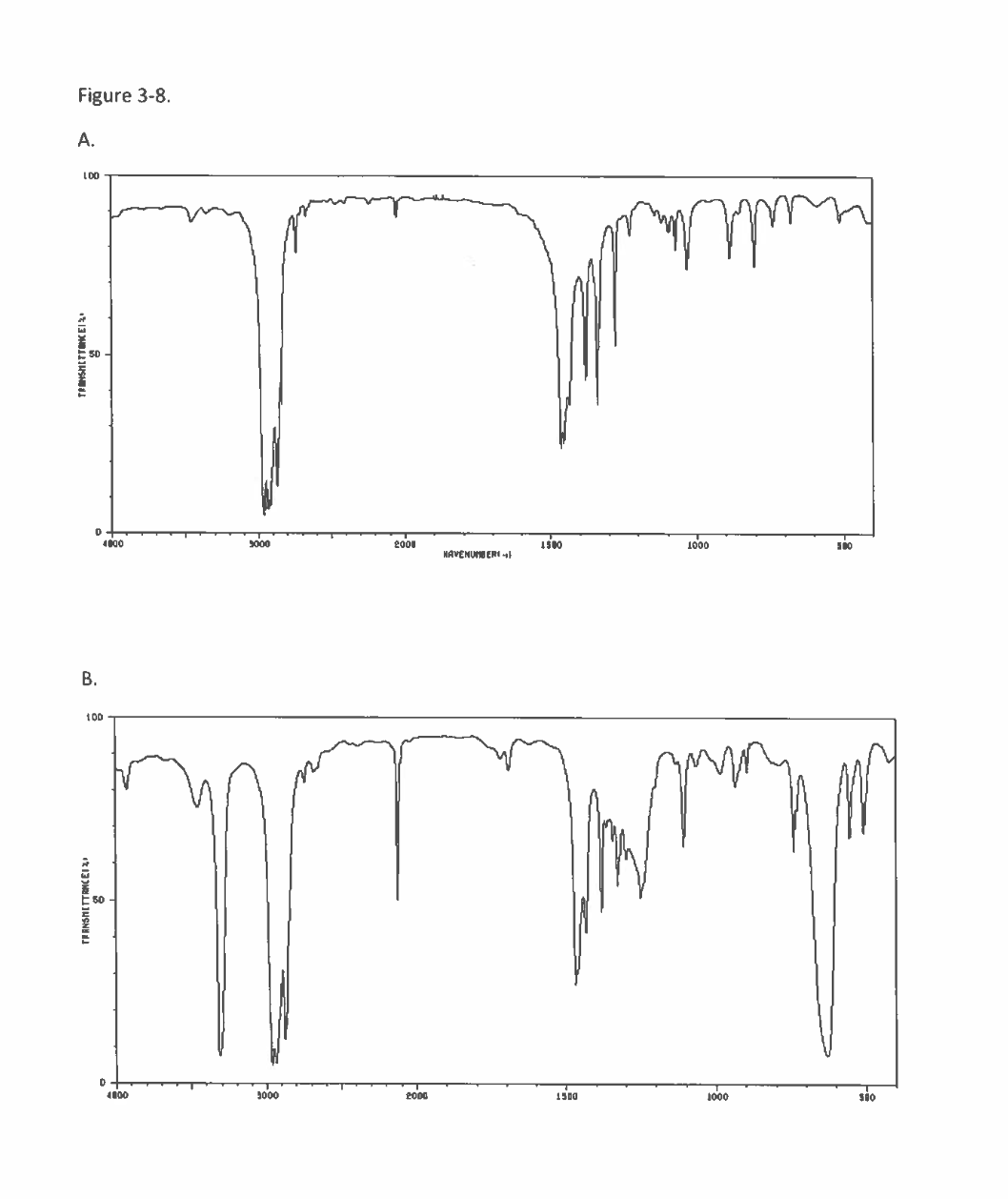
Transcribed Image Text:Figure 3-8.
mm
www
прип
A.
100
TRANSHETTONE
D
B.
4000
100
TRANSMITTANCE
D
4800
3000
3000
2000
2000
HAVENUMBER
15.00
1500
1000
1000
100
300
![See Figure 3-8.
Spectrum A is a [Select]
Spectrum B is
✓ [Select ]
internal alkyne
terminal alkyne](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F34a9c4cb-86d3-4919-ad5f-37407f4c9cd7%2Feb352d80-111e-4cec-83c8-1d2d2bac391d%2Fkj3cufo_processed.png&w=3840&q=75)
Transcribed Image Text:See Figure 3-8.
Spectrum A is a [Select]
Spectrum B is
✓ [Select ]
internal alkyne
terminal alkyne
Expert Solution
Step 1
IR spectroscopy based on the bond strength of different bonds present in a molecule. hence it helps to detect different functional group present in a molecule. As the bond strength increases, the frequency i.e. wavenumber also increases. And the bond strength influenced by different factors like hybridization, electronegativity, atomic mass etc.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

