Syllabus Modules Question 25 Bohr was able to mathematically calculate the wavelength of light in the hydrogen atom due to electronic transitions between energy leve Calculate the wavelength of light in nanometers (nm) associated with the transition from the higher energy level, n = 3 to a low energy leve 28 ALEKS Grades Zoom 1. Tutoring Brainfuse Tutoring Library Services Class Climate Survey Evaluation E Exp 11 G4 Report Only - Google x Question 26 a docs.google.com/document/d/fi0-blz298 Jm9tthGlmK6LmplCfr7 s8gFRjhil24/edit For Aluminum, there ☆ロ の Exp 11 G4 Report Only File Edit View Insert Format Tools Add-ons He O Share a second jump betwe Edit View Insert *a Aア 100% Normal text Cambria 12 ... 12pt v Paragraph jons 2_ HCl a) + ZnC12 (aq) + H2 Laboratory Report Zn Cher A Net lonic Reactions for Doubl. B. Net Reactions of Oxidation R. Observations: Rxn bubbles v C Reactivity of metals: Activa Go Sen Oxidation Half Reaction: Typ haet anch Zinc atoms ar Addess 50'F Cloudy acer | 個司
Syllabus Modules Question 25 Bohr was able to mathematically calculate the wavelength of light in the hydrogen atom due to electronic transitions between energy leve Calculate the wavelength of light in nanometers (nm) associated with the transition from the higher energy level, n = 3 to a low energy leve 28 ALEKS Grades Zoom 1. Tutoring Brainfuse Tutoring Library Services Class Climate Survey Evaluation E Exp 11 G4 Report Only - Google x Question 26 a docs.google.com/document/d/fi0-blz298 Jm9tthGlmK6LmplCfr7 s8gFRjhil24/edit For Aluminum, there ☆ロ の Exp 11 G4 Report Only File Edit View Insert Format Tools Add-ons He O Share a second jump betwe Edit View Insert *a Aア 100% Normal text Cambria 12 ... 12pt v Paragraph jons 2_ HCl a) + ZnC12 (aq) + H2 Laboratory Report Zn Cher A Net lonic Reactions for Doubl. B. Net Reactions of Oxidation R. Observations: Rxn bubbles v C Reactivity of metals: Activa Go Sen Oxidation Half Reaction: Typ haet anch Zinc atoms ar Addess 50'F Cloudy acer | 個司
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter7: The Structure Of Atoms And Periodic Trends
Section: Chapter Questions
Problem 66IL: Spinets are solids with the general formula M2+ (M3+)2O4 (where M2+ and M3+ are metal cations of the...
Related questions
Question
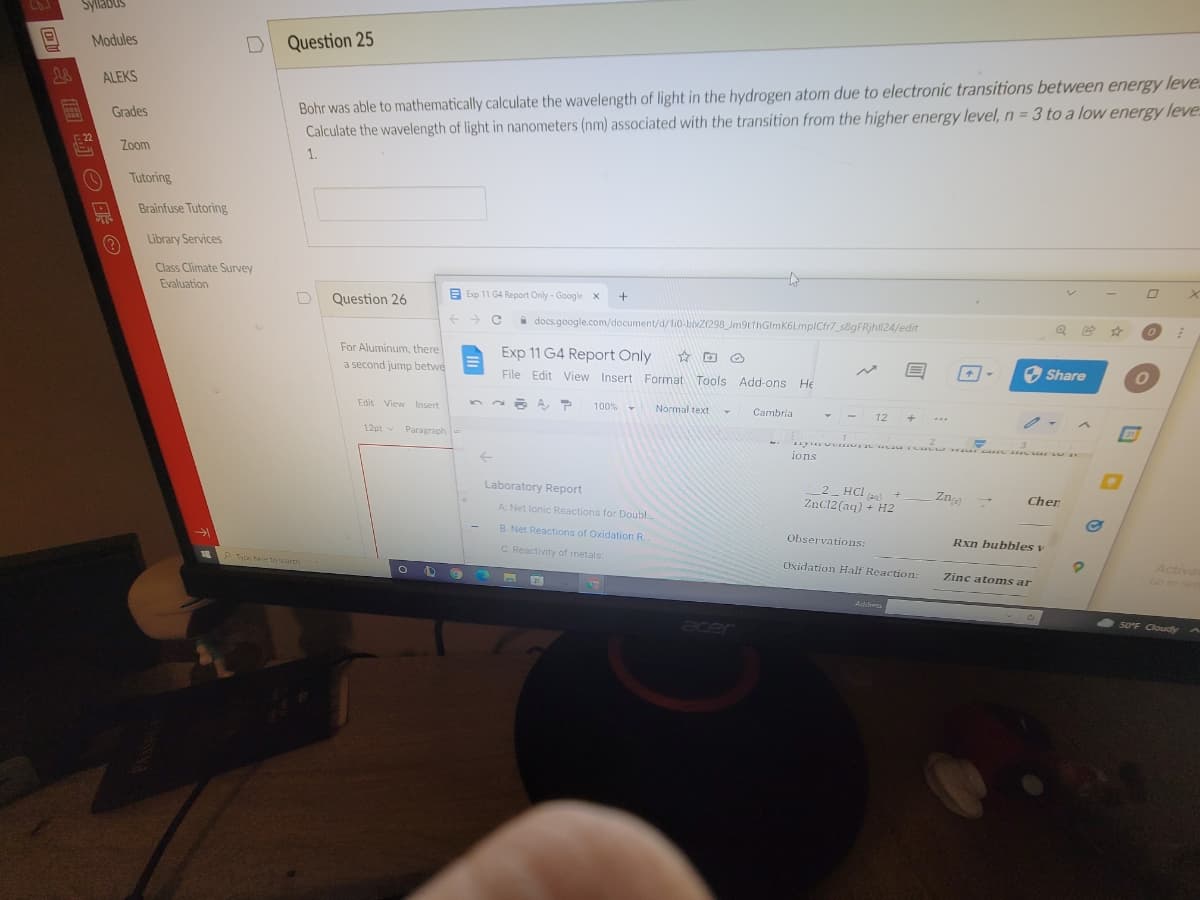
Transcribed Image Text:Syllabus
Modules
Question 25
Bohr was able to mathematically calculate the wavelength of light in the hydrogen atom due to electronic transitions between energy leve
Calculate the wavelength of light in nanometers (nm) associated with the transition from the higher energy level, n= 3 to a low energy leve
28
ALEKS
Grades
Zoom
1.
Tutoring
Brainfuse Tutoring
Library Services
Class Climate Survey
Evaluation
E Exp 11 G4 Report Only - Google x
Question 26
i docs.google.com/document/d/li0-bixz298 Jm9t1hGlmK6LmplCfr7 s8gFRjhll24/edit
For Aluminum, there
a second jump betwe
Exp 11 G4 Report Only
File Edit View Insert Format Tools Add-ons He
O Share
Edit View Insert
100%
Normal text
Cambria
12
...
12pt v Paragraph-
jons
2_ HCl a) +
ZnC12 (aq) + H2
Laboratory Report
Zno
Cher
A Net lonic Reactions for Doubl.
B. Net Reactions of Oxidation R.
Observations:
Rxn bubbles v
C. Reactivity of metals:
Activa
Oxidation Half Reaction:
P Typetee0carch
Zinc atoms ar
GO to Se
Addess
50'F Cloudy
acer
| 個司O联
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning