Table B: Reaction Rate of Na S O/HCI A-Volume of 0.15 M Na S,O, (aq) used / ml. 50.0 40.0 30.0 Beaker # 12345 20.0 10.0 B- Total Volume of volume water/mL. (A+B) /ml 50.0 50.0 50.0 0.0 10.0 20.0 30.0 40.0 50.0 50.0 *Concentratio n of Na,S,0, (aq) /mol L 15 12 Plot two graphs: 1. Time vs concentration of Na S,0, (aq) 2 1/Time vs concentration of Na S,O, (aq) 090 060 030 Time/s 22.5 27.3 35.1 60.0 159.1 Explain the observations (relationship between concentration and time). 1/time/ st Use civicV; to determine the concentration of the diluted sodium thiosulphate solution (beakers 2-5). On how to use this dilution formula, then, please watch: 0444 0366 0285 0167 00629
Table B: Reaction Rate of Na S O/HCI A-Volume of 0.15 M Na S,O, (aq) used / ml. 50.0 40.0 30.0 Beaker # 12345 20.0 10.0 B- Total Volume of volume water/mL. (A+B) /ml 50.0 50.0 50.0 0.0 10.0 20.0 30.0 40.0 50.0 50.0 *Concentratio n of Na,S,0, (aq) /mol L 15 12 Plot two graphs: 1. Time vs concentration of Na S,0, (aq) 2 1/Time vs concentration of Na S,O, (aq) 090 060 030 Time/s 22.5 27.3 35.1 60.0 159.1 Explain the observations (relationship between concentration and time). 1/time/ st Use civicV; to determine the concentration of the diluted sodium thiosulphate solution (beakers 2-5). On how to use this dilution formula, then, please watch: 0444 0366 0285 0167 00629
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.33PAE: The following experimental data were obtained for the reaction of \'I14* and NOf in acidic solution....
Related questions
Question
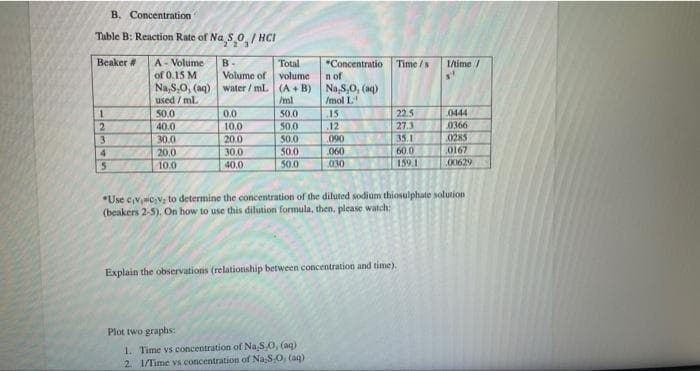
Transcribed Image Text:B. Concentration
Table B: Reaction Rate of Na S O/HCI
Beaker #
12345
A-Volume-
of 0.15 M
Na,S,O, (aq)
used / ml.
50.0
40.0
30.0
20.0
10.0
B-
Volume of
water/mL.
0.0
10.0
20.0
30.0
40.0
Total
volume
(A+B)
/ml
50.0
50,0
50.0
50.0
50.0
*Concentratio
n of
Na,8,0, (aq)
/mol L
15
.12
090
060
030
Plot two graphs:
1. Time vs concentration of Na.S,O, (aq)
2. 1/Time vs concentration of Na S.O, (aq)
Time/s
22.5
27.3
35.1
60.0
159.1
Explain the observations (relationship between concentration and time).
1/time/
0444
0366
*Use civic₂V; to determine the concentration of the diluted sodium thiosulphate solution
(beakers 2-5). On how to use this dilution formula, then, please watch:
0285
0167
00629
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 6 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
