Q: Problem: Would the following transition state lead to a successful reaction? Explain your answer. H…
A:
Q: Show
A: We will write the conversion Of the given reaction
Q: Provide the mechanism for the 1st reaction in this synthesis: КОН
A: We have to draw the mechanism of given reaction
Q: (CH3),CCI ?
A:
Q: For each of the following reactions, draw a complete, detailed mechanism and predict the major…
A: . When aluene react with Bromine in water medium we got addition product . Nucleophite attack…
Q: H2C (SYN) (a) Draw the alcohol that would be required to form the alkyl chloride shown here using…
A: The reaction between alcohol and phosphorous chloride (PCl3) results in chloroalkanes.
Q: Predict the product of the reaction between acetic anhydride and each of the following compounds. If…
A: As here question has multiple subparts, so solve first three subparts. If student want complete…
Q: For each reaction, draw the complete, detailed mechanism and the major organic product. (a) (b)…
A:
Q: Draw a detailed mechanism showing the formation of all products, be sure to draw all intermediate…
A: In the organic conversion, suitable reagent converts the reactant into products. Reagents attacks…
Q: williamson
A: In Williamson etherification Synthesis reagent is used NaOH or NaH ( base ) and solvent is THF .…
Q: Br MeO, THE + NaOMe
A: Welcome to bartleby !
Q: (SYN) How would you synthesize the compound shown here if, as your starting material, you may use…
A: PCC- It is Pyridinium Chloro chromate. It is used as good oxidizing agent. It is used in oxidation…
Q: Draw the complete, detailed mechanism for the following reaction.
A: Base abstracts a hydrogen atom from CH3NO2 and it generates a good nucleophile (-CH2NO2). This…
Q: 1. Draw the complete, detailed mechanism (including curved arrows) for the following reaction…
A: Alkyl halide gives a nucleophilic substitution reaction. In the nucleophilic substitution reaction,…
Q: The phenyl group, C,H, is known to be an ortho/para-directing group. (a) With that in mind, predict…
A: Para product is majored.
Q: OH p-TSOH, C6H8
A: In this reaction, firstly carbocation is generated then ring expansion take place then base abstract…
Q: (SYN) Show how you would carry out the synthesis at the right using the starting material given,…
A: Alkynes- Unsaturated hydrocarbon containing carbon carbon triple bond is known as alkyne. Alkynes…
Q: Topose a synthesis of the aldehyde using ethane and propane as your only sources of carbon atoms.…
A: Since you have posted multiple questions in a session, as per guidelines we are entitled to answer…
Q: A thiophene ring is sufficiently activated that bromination may take place without the presence of a…
A: Treatment of alkenes with Br2 gives vicinal dibromides (1,2-dibromides). The bromines add to…
Q: The crossed aldol reaction shown here can be carried out using a weak base such as pyridine. (a)…
A:
Q: Illustrate Mechanism Nucleophilic Addition—A Two-Step Process ?
A: The nucleophilic addition reaction is a characteristic reaction shown by carbonyl compounds like…
Q: Draw the major product of the following reaction and enter its InChl code in the space provided.…
A: This is a nitration reaction, in this reaction nitronium ion generated from the nitrating mixture…
Q: Draw the detailed mechanism for the following:
A: In the first step- electrocyclic ring opening occur
Q: What alkene yields attached set of oxidative cleavage products ?
A: What alkene yields an attached set of oxidative cleavage products has to be determined.
Q: (SYN) Propose how you would carry out the transformation shown here. Hint: It may take more than a…
A: Given reaction:
Q: ce
A:
Q: Draw a complete, detailed mechanism AND prediet the major organic product for the follawmg trans…
A:
Q: water ?
A: Reaction with water is unimolecular nucleophilic substitution reaction giving enantiomeric products.
Q: Explain Please, Thank You: Which of the following substrates can undergo an E2 step with H2N- as the…
A: The reaction occurs via E2-mechanism only if the hydrogen and leaving group are anti to each other…
Q: Show any reaction that attaches one or two deuterium atom(s) selectively on carbon.
A: Deuterium is an isotope of hydrogen. It is a double standard gas as it has two atoms in one atom. It…
Q: In the acid-catalyzed aromatic alkylation involving 1-methylcyclohexene and benzene, two isomeric…
A:
Q: Draw a reasonable, detailed mechanism that shows this racemization at the a (alpha) carbon. Note:…
A: When the mixture of the stereoisomers (R or S) present in equal proportions, it provides a racemic…
Q: (SYN) In the reaction shown here, the aromatic ring has just one chemically distinct, aromatic H, so…
A: The given reaction is as follows,
Q: НО Draw a full mechanism that illustrates the hydrolysis of the compound above
A: The imine functional group is identified by the presence of a carbon-nitrogen double bond. It is…
Q: Identify the product of the following one-step sequence. Note that this is a reaction that you have…
A:
Q: (SYN) Show how to carry out each of the following syntheses by first converting the alcohol into a…
A: (a) Given reaction is In the above reaction, the alcohol can be converted into sulfonyl chloride bu…
Q: Draw a complete, detailed mechanism for the reaction shown here.
A:
Q: Please help me draw the MAJOR product(s) of the this reaction.
A: It is an example of Friedal craft alkylation reaction
Q: Please draw the major product/products of each of the following reaction
A: We will write mechanism of the reaction and estimate the products of this reaction.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. H3O*, heat P Type here to…
A:
Q: BuLi
A: Organolithium is a reagent that acts as a base or nucleophile in the reaction as per the given…
Q: (SYN) Show how each of the following compounds can be synthesized from an acid chloride and either…
A: Reactions of acid chlorides are,
Q: (SYN) Suggest how you would carry out the synthesis shown here using any reagents necessary. Hint:…
A:
Q: Complete the following reaction and provide the detailed mechanism H*
A:
Q: Circle the molecule that will undergo electophilic aromatic substitution at a faster rate
A: To solve this problem we have to know about the factor which effect the aromatic electrophilic…
Q: Provide a multi-step synthesis of the target structure on the right from the starting structure on…
A: Here we have to synthesize the following given target product from the given starting material by…
Q: Predict the major organic product of this reaction and draw the complete mechanism to form it:…
A:
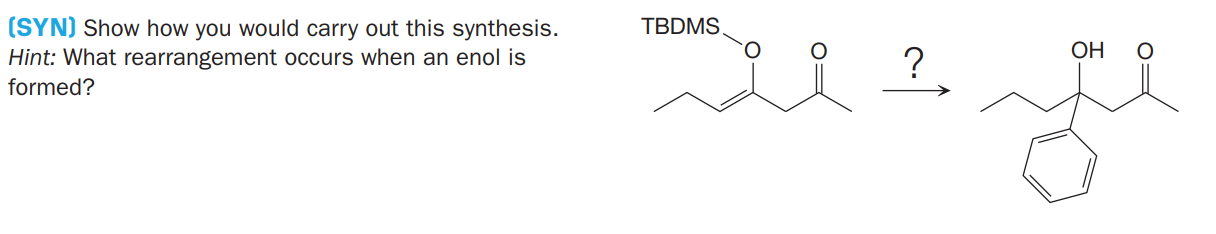
Q: (SYN) Suggest how you would carry out the synthesisshown here using any reagents necessary. Hint:…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Explain Please, Thank You: Which of the following substrates can undergo an E2 step with H2N- as the base? For those that can, draw the curved arrow notation and products.(SYN) In the reaction shown here, the aromatic ring has just one chemically distinct, aromatic H, so a single electrophilic aromatic substitution will lead to just a single product. With this in mind, supply the missing reagentsneeded to carry out the transformation.please fill in the table as per the instruction. Indicate the major enolate, electrophile and product. Make sure to indicate the above mentioned!! Thanks
- (SYN) Show how to carry out this transformation using any reagents necessaryCould you please check if this mechanism is correct and respond constructively any alternate route or reaction? I’m also having hard time on this other problem and I was wondering you could elaborate the steps?Fill out the table per instructions, I have attached two exact same pictures incase one is not visible, make sure to indicate the electrophile, major enolate, product separately and answer all questions. Thanks!
- What is the majpr product resultring from the following series of reactions? The answer is B, please draw a detailed reactions with steps:)) Thanks!!synthesis, please propose a sequence that converts the starting to the target, draw structure of product after each synthetic stepochem help please... What is the major product of the following reaction sequence? (see attached image)
- Please draw a reasonable SN1 mechanism leading to the products shownPlease draw a full mechanism of the reaction. Please do not copy from previously answered question.Help, explain in detail please. Thank you! Did the following overall reaction occur by an SN2, SN1, E2, or E1 mechanism? How do you know? Draw the complete, detailed mechanism to account for the formation of both products.

