Q: What is IUPAC name for the structure with a condensed formula (CH₂)₂CHCH₂COCH, Type your response
A: IUPAC stands for the International Union of Pure and Applied Chemistry. It is a global organization…
Q: Any substance that is an electrolyte must dissolve to form ions when placed in water. Answer: TRUE,…
A: An electrolyte is a substance which when dissolve in water give ions and can conduct electricity.…
Q: An unknown compound has the following chemical formula: PCI where x stands for a whole number.…
A:
Q: An unknown compound has the following chemical formula: NOX where x stands for a whole number.…
A: Given: Chemical formula: NOX X stands for a whole number moles of Oxygen = 8.70 mol moles of…
Q: The equilibrium constant, K, for the following reaction is 1.80E-2 at 698 K 2 HI(g) H₂(g) + 12(g) An…
A: Answer: Value of equilibrium constant is equal to the ratio of molar concentration of products and…
Q: Which of the following is always true about a unimolecular reaction? (A) The activation energy is…
A: The question is based on the concepts of chemical kinetics. unimolecular reaction is the reaction in…
Q: Consider the malate dehydrogenase reaction from the citric acid cycle. Given the listed…
A:
Q: b) Ph CN 1) MeLi 2) NaBH4 3) PBr3 4) Mg 5) CO₂ (dry ice)
A: Given that, a reaction sequence is shown below We have to give the final product of the above…
Q: Indicate, reasoning your answer based on the mechanisms of the reactions involved, which is the best…
A: A compound that contains a double bond between two carbon atoms is known as an alkene. The formation…
Q: Indicate how you would carry out the following syntheses: a) Cis-2-heptene from propyne b) Butanal…
A: The question is based on the concepts of Organic synthesis. we need to synthesize the product based…
Q: Converting aldehyde to hydroxyketone using a-silyloxyketones
A: Answer: The conversion of an aldehyde to a hydroxyketone can be achieved using α-silyloxyketones as…
Q: Tutored Practice Problem 18.2.3 COUNTS TOWARDS GRADE Use the reaction quotient to predict whether a…
A: Given : Volume of Ba(OH)2 = 12 ml Concentration of Ba(OH)2 = 1.10 x 10-4 M Volume of Na2CO3 =…
Q: Based on the data given answer questions. 1,3, and 5 please ?? Question 2 is already answered to…
A: The question is based on the concept of experimental Chemistry. we need to discuss precision and…
Q: Which of the following transport systems allows small molecules such as urea and O₂ to cross the…
A: By the passive diffusion process small molecule cross the cell membrane without expanding energy .…
Q: Draw the product of this series of reactions. 1. 1 equivalent of NaNH₂ 2. CH₂CH₂Br 3. H₂/ Lindlar's…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: 1) What is the formula for lanthanum(III) fluoride? 2) What is the formula for selenium…
A: ANS. Formula of lanthanum(III) fluoride is : LaF3
Q: Which of the following are NOT amphoteric? Select all that apply. PO43- H₂PO4 H₂O OH H3O+ HCO3-…
A:
Q: 1. How do you account for the smell of vinegar when an old bottle of aspi 2. The carbonyls of…
A: The question is based on the concept of IR spectroscopy. We need to account for different…
Q: 5. The equilibrium constant (Kp) for the reaction: N₂O4 (9) #2 NO2 (g) at 25°C is 0.148. A certain…
A: The Gibbs free energy change (∆G) of a reaction and the corresponding reaction quotient (Q) is…
Q: The formation constant* of [M(CN) 6 ]4− is 2.50 × 10 17 , where M is a generic metal. A 0.140 mole…
A: Given , Formation constant of [M(CN)6]4-=2.50×1017
Q: Choose the major product obtained from Diels-Alder reaction of cycloaddition of 1,3-cyclopentadiene…
A: When a conjugated diene reacts with dienophile it forms six member ring as a product
Q: What is the hydroxide ion concentration of a solution prepared by dissolving 0.658 g of Mg(OH)2 in…
A: Given that - Mass of magnesium hydroxide Mg(OH)2 = 0.658 g Volume of solution (of water) = 333 mL…
Q: In classroom demonstration, a teacher pours a supersaturated solution of sodium acetate and water…
A: Given In classroom demonstration, a teacher pours a supersaturated solution of sodium acetate and…
Q: hello i am having difficulty with part a, b, and c A galvanic cell based on the above reaction is…
A: Galvanic cell
Q: 1a) Assuming standard conditions, answer the following questions. (Use the table of Standard…
A: The species with the standard reduction potential greater than H+ would oxidize the H2.
Q: Show how you might synthesize this compound from an alkyl bromide and a nucleophile in an SN2…
A: For the synthesis of the following compound, we have first identify the bond that can be easily…
Q: In which state of matter are the particles least tightly arranged?
A: Given, which state particles are least tight arranged- Basically, three states of matter- Solid,…
Q: Draw a bond-line structure having the molecular formula [C6H12N]+. If applicable, show lone electron…
A: We have to draw the bond line structure of the compound.
Q: If 364 mL of sulfuric acid is diluted to 1.00 L to give a solution that is 1.94 M, what was the…
A:
Q: Consider data from the table of standard reduction potentials for common half-reactions, in your…
A: The anodic half -reaction that would produce a spontaneous reaction of Sn4+ to Sn2+ but not Sn2+ to…
Q: A study of the rate of the reaction represented as 2 A ⟶ B gave the following data: Time (s) 0.0…
A:
Q: How many milliliters of 0.506 M H 2SO 4 would be required to exactly neutralize 891 mL of 0.876 M…
A:
Q: The pKa for Bis-Tris buffer is 6.5. What is the concentration of Bis-Tris Base if the pH is 6.9 and…
A:
Q: Which of the following substances is/are never in a Bronsted-Lowry acid in an aqueous solution? KOH…
A: A Bronsted-Lowry acid is a substance which donates a proton or H+ ion to the other compound
Q: Attempt 3 Calculate the pH of the resulting solution when 25.0 mL of 0.30 M HCl*O_{4} is added to…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: Write the mechanism that explains this reaction. Don't write anything in the box. KOCH,CH, + Sill…
A:
Q: Sam drops a sugar cube into a jar with hot water and a sugar cube into a jar with cold water. Both…
A: Given Sam drops a sugar cube into a jar with hot water and a sugar cube into a jar with cold water.…
Q: =) Complete the following equations ) H₂C. OH H Mario + H₂C OH HO =) H₂C OH OH HO HO CH3 H* CH3 -CH3…
A: sterification is the process of combining an organic acid (RCOOH) with an alcohol (ROH) to form an…
Q: If the concentration of a reactant in a chemical reaction is increased, according to Le Chatelier's…
A: According to Le Chatelier's principle, if a dynamic equilibrium is disturbed by changing the…
Q: How many cubic centimeters of water should be mixed with 25g of 30% (w/w) sulfuric acid and 50g of…
A: Answer: Formula to calculate mass percent of solute is shown below: (w/w)%=mass of solutemass of…
Q: Why is a surfactant used in the making a ferrofluid?
A: Ferrofluids are a unique class of materials that exhibit fascinating magnetic properties and have…
Q: 1 7. Routine analysis of a water sample provides the following concentrations (in M). a) What is the…
A: Ionic strength is a measure of the concentration of ions in a solution and their ability to conduct…
Q: A sealed container holding 0.0255 L of an ideal gas at 0.985 atm and 71 "C is placed into a…
A:
Q: Is it 10 or 3.55
A: 4. Recall the given reaction, CaMgCO32 ↔ Ca2+ + Mg2+ + 2 CO32-at 25 °C…
Q: Which of the following would successfully perform the shown reaction? (A) (B) (C) OH 1) NaH 2) EtBr…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Give detailed Solution with explanation needed
A:
Q: 22-90. Calculate the equilibrium mercury(I) ion con- centration in a solution made by mixing 100.0…
A: Equilibrium concentration of Hg + ion is.
Q: Draw the major product and the mechanism. d. e. f. g. Br Hill میں -CF3 NaOH₂ Br H₂O heat Br Na H₂…
A:
Q: nterpret the CNMR and HNMR
A: 1H-NMR spectroscopy is mainly used for the identification of structure of unknown compound. From the…
Q: Calculate the pH in the titration of 100.0 mL, of 0.20 M HCIO (K = 4.0 x 10^-8) with 0.20 M KOH…
A: Given that, Volume of HClO=100.0mL Concentration of HClO=0.20M Ka=4.0*10-8 Concentration of…
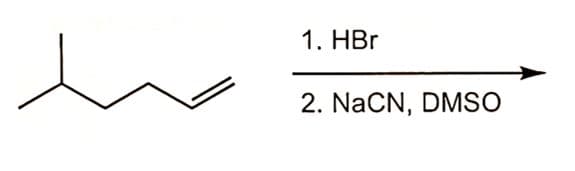
ochem help please...
What is the major product of the following reaction sequence? (see attached image)
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- In one sentence explain what is wrong with them, and rewrite them so that the coefficients are correct. 2 Mg(OH)2(s)+4HCl(aq)->2MgCl2(aq)+4H2O(l)JJ Thomson is credited with the discovery ofZn + 2 HCl ------> ZnCl2 + H2 2) When HCl(aq) is poured over the zinc metal, the liquid begins to fizz with small bubbles. What are the bubbles made of? Combustion Tests of Hydrogen –



