Q: Conductivity of 0.001028 M of acetic acid is 4.95 x 10-5 S cm-1. Calculate the dissociation constant...
A:
Q: When aqueous solutions of nitric acid and barium hydroxide are mixed, the following reaction occurs:...
A: The balanced reaction taking place is given as, => 2 HNO3 + Ba(OH)2 -----> Ba(NO3)2 + 2 H2O Gi...
Q: Determine Ecell for the reaction: 2 Al(aq) +3 Zn2*(aq) → 2 Al3+(aq) + 3 Zn(s). The half reactions ar...
A:
Q: What is the freezing point of 0.260 m urea in water? in C
A: The Depression in freezing point is given as, ∆Tf=(To-T)=kf×mTo=freezing point of pure solvent (for ...
Q: At the end of 2009, global population was about 6.8 billion people. What mass of glucose (C6H12O6) i...
A: Global population at the end of 2009, for whom mass of glucose needed is to be calculated in one yea...
Q: What are the major IR bands, 1H-nmr signals, And the m/z of the following compounds Furan maleic anh...
A: uran is a clear, colorless, flammable liquid cyclic ether with an ethereal odor. Furan is used as an...
Q: A certain gas sample has a volume of 40.00 L at 273 K. At constant pressure, the volume increase o 5...
A: Given, a sample of gas has,initial volume =40.00 Linitial temperature =273 Kfinal volume =50.00 Lwe ...
Q: Write reaction equations for the synthesis of H2SO4, SO2, SO3 and H2SO4. Is all sulphides insoluble...
A: Sulphuric acid, also known by the name of oil of vitrol, is a type of acid made up of sulphur, hydro...
Q: The free energy change for the following reaction at 25 °C, when [Sn2+] = 1.13 M and [Ni2+] = 4.56×1...
A: The Gibb's free energy is given as, ∆G=-nFEcell0 where n is the number of electrons and Ecell0 is th...
Q: A 25 mL of 0.50 M solution of NaOH was diluted to a final volume of 200 mL. What is the new concentr...
A: The given problem relates to stock dilution, so we can use the below relation to calculate the volum...
Q: A medically useful isotope of technetium is produced by beta decay of molybdenum−99.Complete the equ...
A: Given information, A medically useful isotope of technetium is produced by beta decay of Molybde...
Q: A tub of water containing Cr³⁺(aq) is subjected to a current of 1.0 amperes for 60 minutes. How many...
A: An external source of voltage is used to carry out a reaction in case of electrochemical cell. The q...
Q: Al, Al2O3 en Al(OH)3 is amphoteric. Explain what the statement means? Give reaction equations to sup...
A: The amphoteric behavior of oxides can be explained in the term of reactivity toward the acid and bas...
Q: The standard potentials for the reduction of nicotinamide adenine dinucleotide (NAD+) and oxaloaceta...
A: The reactions given are, 1) Oxaloacetate2- (aq) + 2 H+ (aq) + 2 e- -------> Malate2- (aq) ...
Q: Write the beta decay for thorium-230.
A:
Q: A proton is accelerated to 86.2 MeV per particle. What is this energy in kJ/mol?
A: Given : Kinetic energy of proton in MeV = 86.2 MeV Since 1 MeV = 106 eV => Kinetic energy of prot...
Q: The Kb of ammonia is 1.76 × 10-5. The pH of a buffer prepared by combining 45.0 mL of 0.180 M ammoni...
A: pH is a scale used to specify the acidity or basicity of an aqueous solution. A buffer solution is o...
Q: A solution is prepared by diluting 15.0 mL of 13.7-M phosphoric acid solution to a final volume of 2...
A: Welcome to bartleby! Introduction : We have to calculate concentration of new solution .
Q: When the Pb+ concentration is 1.45 x 10 4 M, the observed cell potential at 298 K for an electrochem...
A: Welcome to bartleby ! Introduction : We have to calculate the concentration of [Cr3+]
Q: Predict the magnetic properties and determine the spin-only moment for the following: [Mn(H2O)6]2+...
A:
Q: Tin(II) oxidizes spontaneous to Tn(IV) in air. Show reaction equation for this oxidation
A: Tin (Sn) exists in two oxidation states +2 and +4.
Q: Chemistry Question
A: The molar concentration of all the ions of a salt can be obtained knowing the molarity of the salt w...
Q: In the hydrogen atom, the electron is in its lowest energy state, n=1. The maximum electron energy t...
A: Introduction Avogadro’s number: It is also known as Avogadro’s constant One mole of substance is .6....
Q: [References] What is the standard free-energy change AG at 25°C for the following reaction? H2 (9) +...
A:
Q: The equilibrium constant for a chemical reaction is Kc = 902 at 25°C. What is the standard free ener...
A: We know that, the standard free energy change of the reaction is related to the equilibrium constant...
Q: 1. What is the difference between conductivity of solutions and metallic conductivity?
A: Differences are stated as shown below,
Q: HI has a normal boiling point of –35.4°C, and its DHvapis 21.16 kJ/mol. Calculate the molar entropy ...
A:
Q: please help me in item 4 to 10 in chemistry pls
A: The naming of compounds is done based on the chemical formula written and following some rules,
Q: The 175Pt isotope has a half-life of 2.53 seconds. What percentage of the original quantity of 175Pt...
A: The radio active decay follows first order kinetics. The half life t1/2 and rate constant K of a fir...
Q: In which of the following situations would you expect the rate of reaction between marble (solid cal...
A: The rate at which a chemical reaction proceeds is called rate of reaction The rate of reaction depen...
Q: I am Confused on the first table illustrated on the image
A: Given: Colour λ(nm) Red 656.3 Blue 486.1 Violet 434.1 Violet 410.2
Q: Devise a synthesis of 2E-pentene starting from 1-butene, methanol, and any needed reagents. CH3OH
A: The organic reactions are carried out in many steps and with the help of different reagents.
Q: Chemistry Question
A: Rate law gives the relation between the concentration of the reactants and products. Rate = k[A]a[B]...
Q: When the Hg2+ concentration is 7.38×10-4 M, the observed cell potential at 298K for an electrochemic...
A: The standard reduction potential or reduction potential can be useful in determining the directional...
Q: Suppose you mix án unknown clear llgl ade quid floatS itop of the water as a separate layer. What is...
A: Second option. It is non-polar liquid.
Q: What is the sum of the coefficients of the given chemical reaction when balanced?
A: Given, Chemical Reaction: HCl + O2 ---> Cl2 + H2O First , we need to balance the given Reaction....
Q: Which of the following statements concerning the order of a reaction is / are CORRECT? I. The order ...
A: The sum of the concentration terms on which the rate of reaction actually depends is known as order ...
Q: The standard reduction potentials in volts for Pb2+ and Ag+ are -0.13 V and +0.80V respectively. Cal...
A: It is given that the standard reduction potentials, for the Pb2+ half cell is -0.13 V and of the Ag+...
Q: [References] Calculate the equilibrium constant K. at 25°C from the free-energy change for the follo...
A:
Q: Can i get help with these problem
A:
Q: A binary covalent compound contains 78.3% baron and the rest is hydrogen. Its molar mass is approxim...
A: We have Percentage of Boron (B)=78.3% Percentage of Hydrogen (H)=100-78.3=21.7% Atomic mass of B=10...
Q: For the reaction: Mn(s) + AGNO3(aq)→Ag(s) + Mn(NO3)2(aq) Ag (aq) + e→ Ag(s) E° 0.800 V %3D Mn2+(aq) ...
A: A cell reaction between Mn and Ag+ that give Mn2+ and Ag.
Find activity coefficient.
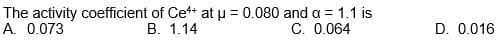
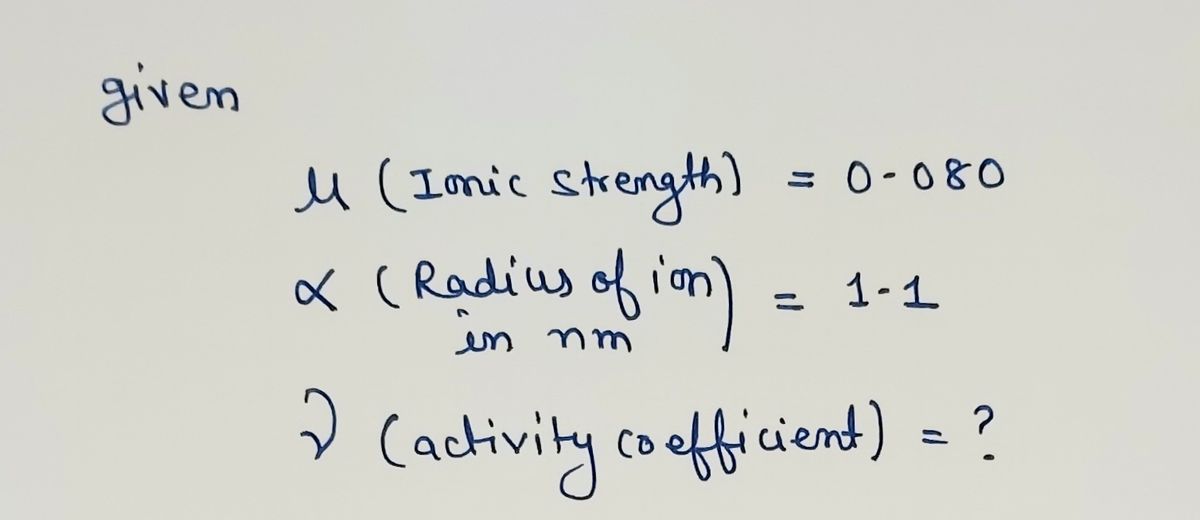
Step by step
Solved in 2 steps with 2 images

- Arrange the gases CO, N2 and CH4 in increasing order of adsorption on the surface of charcoal in a closed vessel. Give reasons also.1.At -185°C and 1 atm ethane (MW = 30.07 g/mol) exists as a solid with a FCC structure. Using the data provided below: E/k=194.1K, bo=0.1794 m3/kmol Determine the diameter of one molecule of ethane (in angstroms). 2. What is the void fraction? 3. Find the density of the solid etahne. 4. Calculate the distance of separation (in m) between two ethane molecules when the potential energy is at a minimum. 5. At the separation distance calculated in part (4), determine the minimum value of the potential energy between two molecules (in J).What is the effect of temperature on molar conductivity?
- relation between melting temperature Tm and maximum crystallization temperature of polymerThe purification of hydrogen gas is possible by diffusion through a thin palladium sheet. Calculate the number of kilograms of hydrogen that pass per hour through a 2.8 mm thick sheet of palladium having and area of 0.72 m^2 at 500 C. Assume a diffusion coefficient of 7.8 x 10^-8 m^2/s, that the concentrations at the high- and low- pressure sides of the plates are 3.4 and 0.58 kg/m^3 of hydrogen per cubic meter of palladium, and that steady-state conditions have been attained.1. "Band structure" of "Hatom chain" and "H mole chain" are drawn as "graphs" and "intermediate process". Describe it in detail.
- Saturatad uater vapour at 60c°has iti pressure decrease to ncrease the volume by 10 % Keeping the temperature constant. To what pressure Should be expands.In detail outline the phenomenon and the cause of phase transitions in polymer systems.Explain pharmaceutical and medical applications of interfacial phenomenon. Quote the applications with suitable examples.
- Estimate the mole fraction of Schottky and Frenkel defects in NaCl crystal at 1000K. The energies of the formation of these defects are 2eV and 3eV respectively.Calculate the change in H when one mole of water is heated from 263 to 283 K given the molar capacities inJ.K-1 , Cp(ice) = 2.09 + 0.126T, Cp(water)=75.3, and change in enthalpy at formation is 6000Jmol-1. P=101.32 kPaPolyethylene glycol 400 (PEG400) is a nonvolatile liquid polymer, which when mixed with water canbe used as a base for parenteral formulations. The designation “400” means that the molecular weightof PEG molecules is approximately 400 g/mol. Preparation with water (ΔHv = 40.7 kJ/mol) requires that the two be mixed at 56 oC, at which the equilibrium vapor pressure of pure water is 16.5 kPa. a. If 105 g of PEG400 are added to 500 g of water at 56 oC, what will the boiling temperature of the solution be?

