The Boltzmann distribution is given by N₁ N₁ = =e = e Assume you have a system with two energy levels. If one in a thousand particles is in the excited state at 45.0 °C and 1.2 bar what is the energy difference between the levels (in kJ/mol)?
The Boltzmann distribution is given by N₁ N₁ = =e = e Assume you have a system with two energy levels. If one in a thousand particles is in the excited state at 45.0 °C and 1.2 bar what is the energy difference between the levels (in kJ/mol)?
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.65E: Determine an expression for V/T p, n in terms of and . Does the sign on the expression make sense...
Related questions
Question
5
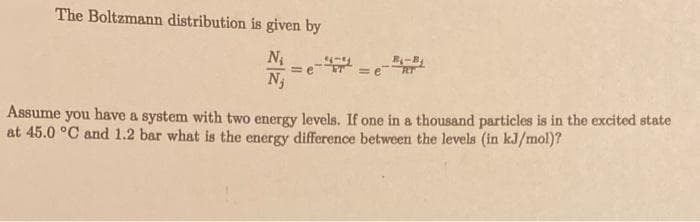
Transcribed Image Text:The Boltzmann distribution is given by
N₁
N₁
=
=e
=e
₁-B₁
Assume you have a system with two energy levels. If one in a thousand particles is in the excited state
at 45.0 °C and 1.2 bar what is the energy difference between the levels (in kJ/mol)?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,