Q: Which alcohol undergoes dehydration fastest ?
A: Dehydration of alcohol is a process in which the alcohols generate alkene upon reaction with acid…
Q: Rank the following compounds in order of increasing boiling point: CH3CH2CH(CH3)2, CH,CH,CO,H, and…
A: The boiling point of organic compounds increases if the molecule able to form Hydrogen bonds.…
Q: 2. Rank the compounds below in order of increasing boiling point -NH2
A: 2. Given compounds,
Q: Rank the following compounds in order of increasing strength of intermolecular forces:…
A: The forces which provide interaction including force of repulsion and attraction acting between the…
Q: Determining Relative Boiling Points Which compound in each pair has the higher boiling point? Which…
A: Boiling point : The temperature where liquid changes into vapor or gas is called boiling point . If…
Q: Rank the following compounds in order of increasing boiling point:
A: The given compounds are To find: Increasing order of boiling point
Q: ) Rank the following in order of increasing boiling point. он но, CH3 A B C Depk the follewin g in e…
A: Boiling point is defined as the amount of heat required to vaporize a liquid. this depends on…
Q: Which compound in each pair has the higher boiling point?
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Rank the following compounds in order of increasing strength of intermolecular forces:…
A: Solution- weakest-1. Pentane - No polar group present2 . Butanal - C=O group little…
Q: Which of the preceding compounds forms hydrogen bonds with a solvent such as ethanol?
A: ANSWERE IS GIVEN BELOW :
Q: Which of the following compounds has the lowest boiling point? Select one: a. CH3CH2CH2CH2F…
A:
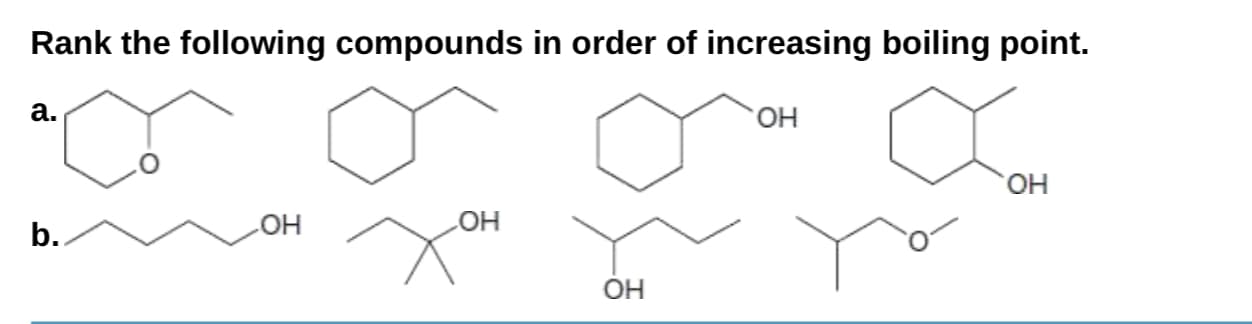
Q: Rank the following compounds in order of increasing boiling point
A: The increasing orders of boiling point of given compounds are as follows:
Q: A E
A: Boiling point depends directly on the surface area. Increase in surface area increases the boiling…
Q: Arrange the following compounds in order of increasing boiling point i.e. start with the least. Use…
A: As we know, boiling point of compound is also depends on hydrogen bonding. As hydrogen bonding…
Q: Rank the compounds in order of increasing boiling point. А. Br В. С. HO. D. HO
A: Boiling point of any molecule depends on 2 things ( in the same priority order as they are…
Q: Rank A–C in order of increasing melting point.
A: The temperature at which the solid phase converted into liquid phase is known as melting point.
Q: Why is the boiling point of propane-1,3-diol (HOCH2CH2CH2OH) higher than the boiling point of…
A: Greater the intermolecular forces, higher the boiling point. 1,2-propanediol and 1,3-propane diol…
Q: Arrange the compounds in the order of increasing boiling point (lowest first)
A: Boiling point depends upon types of interaction between molecules Types Intermolecular force of…
Q: ОН OH OH NH2 Но OH OH
A:
Q: 3. Rank the compounds in the following group in order of increasing boiling point from left to…
A: Since you have asked multiple questions, we will solve the first question for you as per our…
Q: Explain the propertis of ethers that muke them a very weful as menstruum.
A: The main properties of ether because of which it acts as menstruum i.e a solvent are
Q: Write the letter of the following compound(s) that fit the criteria
A: The molecule which will have H present in the molecule connected to the high electronegative atoms…
Q: Rank the following compounds in order of increasing boiling point. The first compound in your rankir…
A: In the given compounds we have different functional groups. So order of boiling point can be…
Q: 17) Which of the following compounds has the lowest boiling point? Select one: a.…
A: To find the compound having the lowest boiling point.
Q: Fruits havé pléäsant odors because of the presence of C. ethyl D. aldehyde A ethers B. esters
A: An ester has an OR group attached to the carbon atom of a carbonyl group. Esters of aromatic acids…
Q: List the following compounds from highest boiling to lowest boiling: Rank the compounds from highest…
A: The factors that affect the boiling point of a compound are, Molecular mass Hydrogen bonding Polar…
Q: Which structure has the highest boiling point? A or B A. Ethers B. Ketones O A. OB.
A: 18. Ketones has higher polarity as compared to ethers. Ethers are of the general form R-O-R' and…
Q: Rank the following compounds in decreasing order of boiling points (highest to lowest): HO II II
A: The boiling point increases by increasing the carbon atoms present in the chain. But in the given…
Q: Arrange the following compounds in order of increasing boiling point.
A: To compare the boiling point, first we need to identify the compounds. - It is a 8 carbon…
Q: Rank the following organic compounds in order of decreasing their boiling points: CH3CH2COOH…
A: Amides are form more number of hydrogen bonds than carboxylic acids, so amides has more boiling…
Q: (a) Which of the following molecules can hydrogen bond to another molecule like itself? (b) Which of…
A: a) The molecules which will have H attached to high electronegative elements like O, N, Cl, and F…
Q: Arrange the compounds in order of increasing boiling point. HO. А В C D
A: Greater is the effective molecular weight and hence, greater is the intermolecular force of a…
Q: Rank the attached compounds in order of increasing boiling point ?
A: Applying concept of hydrogen bonding and vonder Waals interaction.
Q: Arrange the follwing comounds according to their boiling points (lowest to highest) N-pentanol…
A: 1)2-Butanol Boiling point: 99 °C 2) 2-Methyl-1-butanol Boiling point: 129 °C 3)…
Q: Rank the given compounds in increasing order based on the melting point
A: The temperature at which a pure substance's solid and liquid phases can coexist in equilibrium is…
Q: List the following compounds in order of increasing boiling point. H2, CH3OH, H₂S, CH4 A. CH3OH <H₂S…
A:
Q: Rank the compounds in each group in order of increasing boiling point.
A: The order of increasing boiling point has to be given.
Q: Rank the following compounds in order of increasing (lowest to highest) boiling point HO, II III…
A: First question : ( II ) compound is nonpolar and nonpolar compound have weak intermolecular forces…
Q: Rank the following compounds in order of increasing boiling point.
A: The least temperature needed in transformation from solid/liquid to gaseous form is referred as…
Q: Arrange the compounds in order of increasing boiling point.
A: The order depends on the ability to form H-bonding and hence the boiling. Cyclohexylamine can form…
Q: X tetracyanoethylene (A) Dipole-dipole interactions SEG London dispersion forces -0=1 -3=1…
A: The attraction forces between the different molecules are known as intermolecular forces. In general…
Q: Which compound in each pair has the higher boiling point?
A: Boiling points: we compare Boiling points on the basis of the following points: 1) force of…
Q: Which compound has the highest boiling point? 1. CH3CH2-OH 2. CH3CH2-F 3. CH3CH2CH3
A:
Q: arrange the compounds in each set in order of decreasing solubility in water. Q. Ethanol, butane,…
A: Interpreting given different compounds:
Q: Which of the following compounds has a lower boiling points? Provide the main reason for your…
A: The two compounds given are,
Q: Consider the three compounds A -C: OMe CH,OH C b.p. 118°C b.p. 57°C b.p. 65 C (i) (ii) Explain why…
A:
Q: Rank the following compounds in order of increasing boiling point:
A:
Q: Which compound in each pair has the higher boiling point?
A: 1) Since the molar mass of both the molecules is same Hence the boiling point will be proportional…
Q: Rank the following compounds in order of increasing boiling point. Which compound is the most water…
A: The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals…
Q: Arrange the compounds in each set in the order of increasing boiling point.
A: The boiling point of the organic molecule depends on three criteria: The relative strength of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


