The concentration of a certain sodium hydroxide solution was determined by using the solution to titrate a sample of potassium hydrogen phthalate (abbreviated as KHP). KHP is an acid with one acidic hydrogen and a molar mass of 204.22 g/mol. In the titration, 48.82 mL of the sodium hydroxide solution was required to react with 0.7540 g KHP. Calculate the molarity of the sodium hydroxide. Molarity = ___________ M
The concentration of a certain sodium hydroxide solution was determined by using the solution to titrate a sample of potassium hydrogen phthalate (abbreviated as KHP). KHP is an acid with one acidic hydrogen and a molar mass of 204.22 g/mol. In the titration, 48.82 mL of the sodium hydroxide solution was required to react with 0.7540 g KHP. Calculate the molarity of the sodium hydroxide. Molarity = ___________ M
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter4: Types Of Chemical Reactions And Solution Stoichiometry
Section: Chapter Questions
Problem 84E: The concentration of a certain sodium hydroxide solution was determined by using the solution to...
Related questions
Question
The concentration of a certain sodium hydroxide solution was determined by using the solution to titrate a sample of potassium hydrogen phthalate (abbreviated as KHP). KHP is an acid with one acidic hydrogen and a molar mass of 204.22 g/mol. In the titration, 48.82 mL of the sodium hydroxide solution was required to react with 0.7540 g KHP. Calculate the molarity of the sodium hydroxide.
Molarity = ___________ M
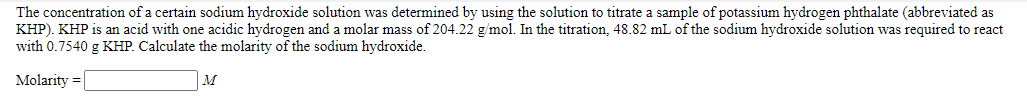
Transcribed Image Text:The concentration of a certain sodium hydroxide solution was determined by using the solution to titrate a sample of potassium hydrogen phthalate (abbreviated as
KHP). KHP is an acid with one acidic hydrogen and a molar mass of 204.22 g/mol. In the titration, 48.82 mL of the sodium hydroxide solution was required to react
with 0.7540 g KHP. Calculate the molarity of the sodium hydroxide.
Molarity =
M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning