Q: Should the final resonance structure be included in the radical resonance? Or is the first and fifth...
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure...
Q: Write the balanced, overall reaction for the situation in which acetate is the donor and carbon sour...
A:
Q: Here is a graph of the molarity of ammonia (NH,) in a reaction vessel during a certain chemical reac...
A: The concentration v/s time curve of NH3 is given as,
Q: A study of the rate of dimerization of CHs gave the data shown in the table, Time (s) 1400. 2800. 42...
A:
Q: A closed cylinder contains 1.0 mole of ideal monatomic gas at 300 o C and 4.0 bar. Insulating the cy...
A: Answer: To change the state of a matter, a thermodynamic process is applied on the system and depedi...
Q: How many grams of PbBr2 will precipitate when excess NH,Br solution is added to 58.0 mL of 0.734 M P...
A: We have to predict mass of lead bromide formed.
Q: MOH base solution is 0.02 M with an ionization degree of 0.02. The pH of the base solution is
A:
Q: Which of the following molecule (s) will give a positive FeCl3 test? Why? OH HO HO НО Но A B Salicyl...
A:
Q: (Q76) Propane combustion results in the production of carbon dioxide and water vapor according to th...
A: We have to predict the rate of reaction.
Q: Consider the sealed containers (same fluid and on the same ground level) below with heights given in...
A: As we descent into Ideal liquid pressure increases linearly with depth. So, pressure inside liquid a...
Q: R - NH2 NH. HO NH DCC,THF NH RT, N2 (atm.) Вос `NH2 Вос `NH2 6(b-e) R= -CH,CH(CH,),.(2b) -CH,Ph,(2e)...
A: The reaction involves deprotection of amine group and results in formation of desired heterocyclic c...
Q: Decide if the following set of quantum numbers is permissible. If not, indicate which value is wrong...
A:
Q: 7. What needs to be done to make the following reaction proceed? но OH A) Heat the reaction. B) Add ...
A:
Q: For the following reaction K = 30.0 2A(aq) -> B(aq) + C(aq) The initial concentration of A is 1.7 M ...
A:
Q: Here is a graph of the molarity of hydrogen peroxide (H,0,) in a reaction vessel during a certain ch...
A: The concentration v/s time curve of H2O2 is given as,
Q: The separation of uranium and plutonium by TBP is the key aspect of nuclear fuel reprocessing. If th...
A: A question based on concentration terms that is to be accomplished.
Q: The solution from which a precipitate was formed. a. Mother liqour b. Titrant c. Saturated solution...
A: Mother liquor is defined as the solution which is left out after a certain component has been remove...
Q: 67. What is the pH at which 0.01 M Co2 ions in solution just precipitate as Co(OH), ? Kp of Co(OH), ...
A: We have to find the pH at which Co+2 ion in solution precipitates
Q: Are side products/by products the same thing as minor products in a chemical reaction?
A: No side products/by products are not the same thing as minor products in a chemical reaction.
Q: Based on the following boiling point information, what compound has the strongest intermolecular for...
A:
Q: What volume in L of a 0.320 M Mg(NO,), solution contains 45.0 g of Mg(NO), ? g Mg(NO,), g Mg(NO,), 4...
A:
Q: What is the structure of the conjugate acid for the reaction shown below? OK NH3 NH C OB OA
A: Conjugate acid if formed by the addition of a proton to the base.
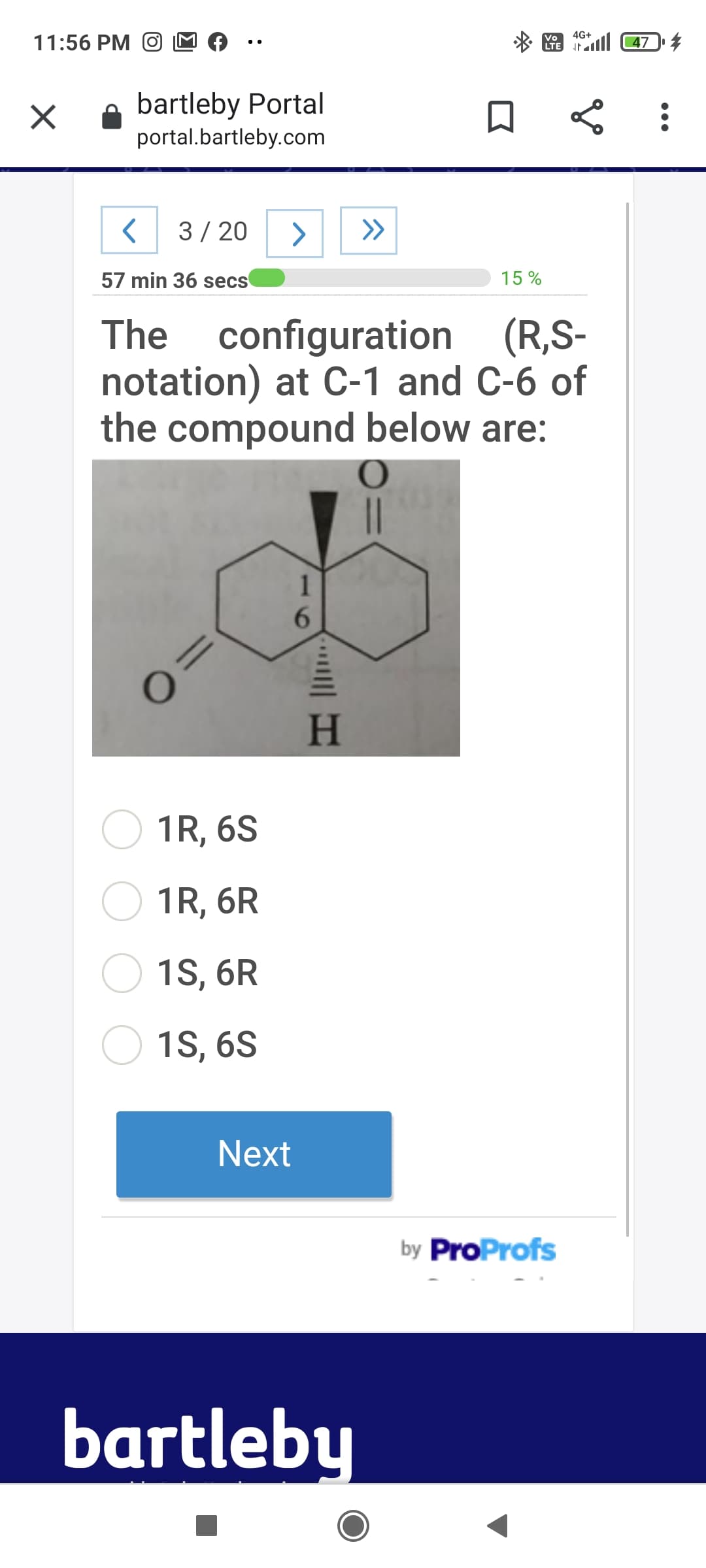
Q: 2. Below is the structure of a newly approved anticancer drug Nubequa. N. NH a. How many stereogenic...
A: Stereogenic center :- The center in molecule (carbon atom) which gives different stereoisomers on th...
Q: Calculate the density of air at atmospheric pressure at sea level and at 15°C, and at a pressure of ...
A:
Q: 74. What is the concentration of Ba" when BaF, (Kp = 1.0 x 10) begins to precipitate from a ds. solu...
A: 74. The solubility product ( Ksp ) of a sparingly soluble salt is defined as the product of the mola...
Q: "green" oxidation The above reaction is one of example of environmental benign synthesis which does ...
A: Green synthesis is an environmentally benign synthesis which does not involve organic solvent and to...
Q: A pH of -1.0 is an H+ concentration of 10M not possible an H+ concentration of 1M an H+ concentratio...
A: Given: pH = -1.0.
Q: with sides es of the
A: To find the Electric potential for the given cube.
Q: ETHANOL Sp.Gr.- 0.791 A cylindrical unknown solid material with a diameter of 10cm and a height of 3...
A: Given: Height of unknown cylinder = 30 cm = 0.3 m Diameter of unknown cylinder = 10 cm = 0.1 m We ...
Q: In the laboratory you dilute 5.20 mL of a concentrated 6.00 M hydroiodic acid solution to a total vo...
A: The Concentration of the dilute solution can be calculated as, M1V1 = M2V2 where, M1 = Initial conc...
Q: If you begin an exercise program and burn 5 Calories per minute by waking, how many hours of waking ...
A: Total weight to loose = 3 pounds Required calories to burn to loose 1 pound = 3500 cal Required calo...
Q: The scattering of sunlight by the mixture of gas molecules in air results in the blue color of the s...
A:
Q: Please draw out the 2 reaction schemes: Ethyl diacetoacetate + hydrazine = pyrazole Ethyl diacetoace...
A:
Q: If the K, of a monoprotic weak acid is 1.8 x 10-6, what is the pH of a 0.35 M solution of this acid?...
A:
Q: Decide if the following set of quantum numbers is permissible. If not, indicate which value is wrong...
A:
Q: 1-Bromobutane was hydrolysed by an aqueous sodium hydroxide (NaOH) solution. CH3CH2CH2C...
A: At first, we need to plot the concentration versus time.
Q: Can anyone please help me with to solve this problem?
A:
Q: In the laboratory you dilute 2.56 mL of a concentrated 12.0 M hydrochloric acid solution to a total ...
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute...
Q: A bottle of NaCl reagent kept in a school lab stockroom was inadvertently contaminated with naphthal...
A: Naphthalene is a substance which can convert directly from its solid state into vapor on heating whe...
Q: Given that K for HF is 6.3 × 10-4 at 25 °C, what is the value of K, for F at 25 °C? K, = Given that ...
A:
Q: Calculate the freezing point of a 10.00 m aqueous solution of ethylene glycol. Freezing point consta...
A:
Q: The equilibrium constant, Kc, for the following reaction is 5.10×10-6 at 548 K. NH4Cl(s) NH3(g) + H...
A:
Q: Predict the shape (molecular geometry) of the following molecules or ions by filling up the table be...
A:
Q: Give 5 techniques that will ensure successful purification of an organic compound by recrystallizati...
A: A question based on analytical separations that is to be accomplished.
Q: 11. A 0.8000-g sample of iron ore containing 21.00% Fe is analyzed gravimetrically. If the final pre...
A:
Q: As much as 8 grams of NaOH (Mr = 40) is dissolved in water until the volume of the solution becomes ...
A:
Q: The crystal structure of lead is FCC and its density is 11.36Mg/m^3. What is its lattice parameter a...
A: The FCC unit cell is the simplest repeating unit in a cubic close-packed structure which consists of...
Q: created Is H2O, being created or destroyed by the chemical reaction? destroyed neither created nor d...
A: The concentration v/s time curve of H2O2 is given as,
Q: OH NH2 Неpt + CH3SO,CI + CH3(CH2)NHET H3C (a) (b) (c) (d) (c) SOCI2 (a) + (b) H3PO4 OH Et Неpt (d) (...
A:
Q: How long will it take for the concentration of A to decrease from 1.400 M to 0.205 for the zero orde...
A:
Step by step
Solved in 2 steps with 1 images

- At constant pressure and 25 C, what is the Delta rH for the following reaction: 2C2H6 (g) + 7O2(g) --> 4CO2(g) + H2O(l) if the complete consumption of 12.5g of C2H6 liberates 752.3 kJ of heat energy?Hexanoic acid was added to an immiscible biphasic solvent sysem, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D1) of hexanoic acid in CCl4 with respect to water.Please asnwer subparts 7,2,3
- What is ΔHsys for a reaction at 51.4 °C with ΔSsurr = 749 J mol-1 K-1 ? Express your answer in kJ mol-1 to at least two significant figures.CH.8 #8 - Please fill in the blank, cirlce your answer for thumbs up!Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4
- Calculate delt g for Pbl2(s) + Pb2+(aq) +2I-(aq) T= 25C Kp= 8.7x10-9Ksp for Co(OH)2 is 5.92x10^-15Phenol reacts with three equivalents of bromine in CCl4 (in the dark) to give a product of formula C6H3OBr3. When thisproduct is added to bromine water, a yellow solid of molecular formula C6H2OBr4 precipitates out of the solution. The IRspectrum of the yellow precipitate shows a strong absorption (much like that of a quinone) around 1680 cm-1. Proposestructures for the two products

