A closed cylinder contains 1.0 mole of ideal monatomic gas at 300 o C and 4.0 bar. Insulating the cylinder so that there is no heat exchange with the surroundings, the gas is expanded to 200 o C and 1.0 bar producing 1247 J of work. a) Is this process reversible or irreversible? b) What is the change in enthalpy (∆H) of the gas? c) What is the change in internal energy (∆U) of the gas?
A closed cylinder contains 1.0 mole of ideal monatomic gas at 300 o C and 4.0 bar. Insulating the cylinder so that there is no heat exchange with the surroundings, the gas is expanded to 200 o C and 1.0 bar producing 1247 J of work. a) Is this process reversible or irreversible? b) What is the change in enthalpy (∆H) of the gas? c) What is the change in internal energy (∆U) of the gas?
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.16QAP
Related questions
Question
A closed cylinder contains 1.0 mole of ideal monatomic gas at 300 o
C and 4.0 bar.
Insulating the cylinder so that there is no heat exchange with the surroundings, the gas is
expanded to 200 o
C and 1.0 bar producing 1247 J of work.
a) Is this process reversible or irreversible?
b) What is the change in enthalpy (∆H) of the gas?
c) What is the change in internal energy (∆U) of the
gas?
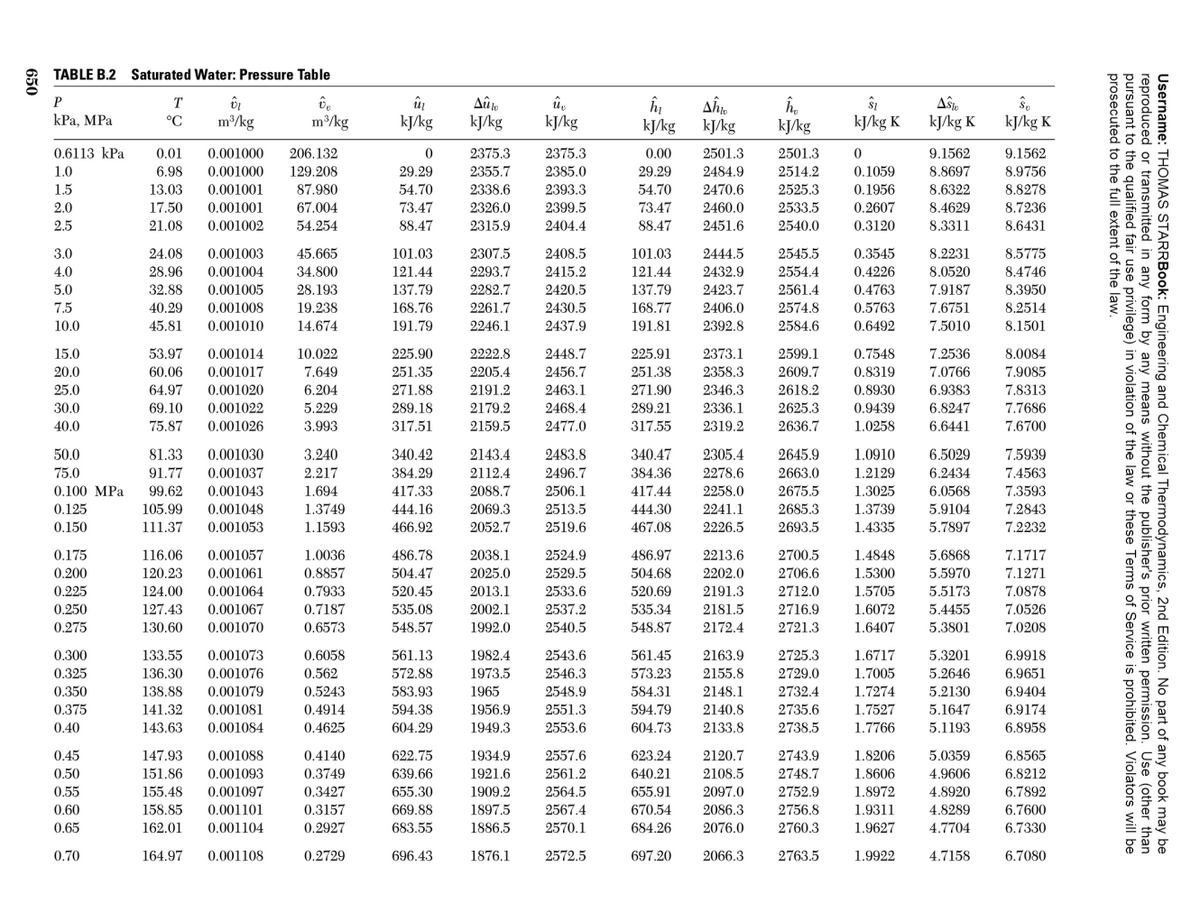
Transcribed Image Text:2 TABLE B.2 Saturated Water: Pressure Table
P
û,
kJ/kg
Aŝte
kJ/kg K
T
КРа, МPа
°C
m/kg
m/kg
kJ/kg
kJ/kg
kJ/kg
kJ/kg
kJ/kg
kJ/kg K
kJ/kg K
0.6113 kPa
0.01
0.001000
206.132
2375.3
2375.3
0.00
2501.3
2501.3
9.1562
9.1562
129.208
87.980
29.29
54.70
8.9756
8.8278
8.7236
1.0
6.98
0.001000
29.29
2355.7
2385.0
2484.9
2514.2
0.1059
8.8697
2393.3
2399.5
2470.6
2460.0
1.5
13.03
0.001001
54.70
2338.6
2525.3
0.1956
8.6322
2.0
17.50
0.001001
67.004
73.47
2326.0
73.47
2533.5
0.2607
8.4629
2.5
21.08
0.001002
54.254
88.47
2315.9
2404.4
88.47
2451.6
2540.0
0.3120
8.3311
8.6431
8.2231
8.0520
3.0
24.08
0.001003
45.665
101.03
2307.5
2408.5
101.03
2444.5
2545.5
0.3545
8.5775
4.0
28.96
0.001004
34.800
121.44
2293.7
2415.2
121.44
2432.9
2554.4
0.4226
8.4746
2561.4
28.193
19.238
14.674
2420.5
2430.5
5.0
32.88
0.001005
137.79
2282.7
137.79
2423.7
0.4763
7.9187
8.3950
7.5
40.29
0.001008
168.76
2261.7
168.77
2406.0
2574.8
0.5763
7.6751
8.2514
10.0
45.81
0.001010
191.79
2246.1
2437.9
191.81
2392.8
2584.6
0.6492
7.5010
8.1501
15.0
53.97
0.001014
10.022
225.90
2222.8
2448.7
225.91
2373.1
2599.1
0.7548
7.2536
8.0084
20.0
60.06
0.001017
7.649
251.35
2205.4
2456.7
251.38
2358.3
2609.7
0.8319
7.0766
7.9085
25.0
64.97
0.001020
6.204
271.88
2191.2
2463.1
271.90
2346.3
2618.2
0.8930
6.9383
7.8313
30.0
69.10
0.001022
5.229
289.18
2179.2
2468.4
289.21
2336.1
2625.3
0.9439
6.8247
7.7686
40.0
75.87
0.001026
3.993
317.51
2159.5
2477.0
317.55
2319.2
2636.7
1.0258
6.6441
7.6700
50.0
81.33
0.001030
3.240
340.42
2143.4
2483.8
340.47
2305.4
2645.9
1.0910
6.5029
7.5939
384.29
417.33
75.0
91.77
0.001037
2.217
2112.4
2496.7
384.36
2278.6
2663.0
1.2129
6.2434
7.4563
0.100 MPa
99.62
0.001043
1.694
2088.7
2506.1
417.44
2258.0
2675.5
1.3025
6.0568
7.3593
0.125
105.99
0.001048
1.3749
444.16
2069.3
2513.5
444.30
2241.1
2685.3
1.3739
5.9104
7.2843
0.150
111.37
0.001053
1.1593
466.92
2052.7
2519.6
467.08
2226.5
2693.5
1.4335
5.7897
7.2232
2700.5
2706.6
0.175
116.06
0.001057
1.0036
486.78
2038.1
2524.9
486.97
2213.6
1.4848
5.6868
7.1717
0,200
120.23
0.001061
0.8857
504.47
2025.0
2529.5
504.68
2202.0
1.5300
5.5970
7.1271
520.45
2712.0
2716.9
0.225
124.00
0.001064
0.7933
2013.1
2533.6
520.69
2191.3
1.5705
5.5173
7.0878
0.250
127.43
0.001067
0.7187
535.08
2002.1
2537.2
535.34
2181.5
1.6072
5.4455
7.0526
0.275
130.60
0.001070
0.6573
548.57
1992.0
2540.5
548.87
2172.4
2721.3
1.6407
5.3801
7.0208
0.300
133.55
0.001073
0.6058
561.13
1982.4
2543.6
561.45
2163.9
2725.3
1.6717
5.3201
6.9918
572.88
1973.5
1965
0.325
136.30
0.001076
0.562
2546.3
573.23
2155.8
2729.0
1.7005
5.2646
6.9651
0.350
138.88
0.001079
0.5243
583.93
2548.9
584.31
2148.1
2732.4
1.7274
5.2130
6.9404
0.375
141.32
0.001081
0.4914
594.38
1956.9
2551.3
594.79
2140.8
2735.6
1.7527
5.1647
6.9174
0.40
143.63
0.001084
0.4625
604.29
1949.3
2553.6
604.73
2133.8
2738.5
1.7766
5.1193
6.8958
0.45
147.93
0.001088
0.4140
622.75
1934.9
2557.6
623.24
2120.7
2743.9
1.8206
5.0359
6.8565
0,50
151.86
0.001093
0.3749
639.66
1921.6
2561.2
640.21
2108.5
2748.7
1.8606
4.9606
6.8212
0.55
155.48
0.001097
0.3427
655.30
1909.2
2564.5
655.91
2097.0
2752.9
1.8972
4.8920
6.7892
669.88
683.55
1897.5
1886.5
670.54
684.26
4.8289
4.7704
0.60
158.85
0.001101
0.3157
2567.4
2086.3
2756.8
1.9311
6.7600
0.65
162.01
0.001104
0.2927
2570.1
2076.0
2760.3
1.9627
6.7330
0.70
164.97
0.001108
0.2729
696.43
1876.1
2572.5
697.20
2066.3
2763.5
1.9922
4.7158
6.7080
Username: THOMAS STARRBook: Engineering and Chemical Thermodynamics, 2nd Edition. No part of any book may be
reproduced or transmitted in any fe
pursuant to the qualified fair use privilege) in violation of the law or these Terms of Service is prohibited. Violators will be
prosecuted to the full extent of the law.
y form by anv
ny means without the publisher's prior written permission. Use (other than
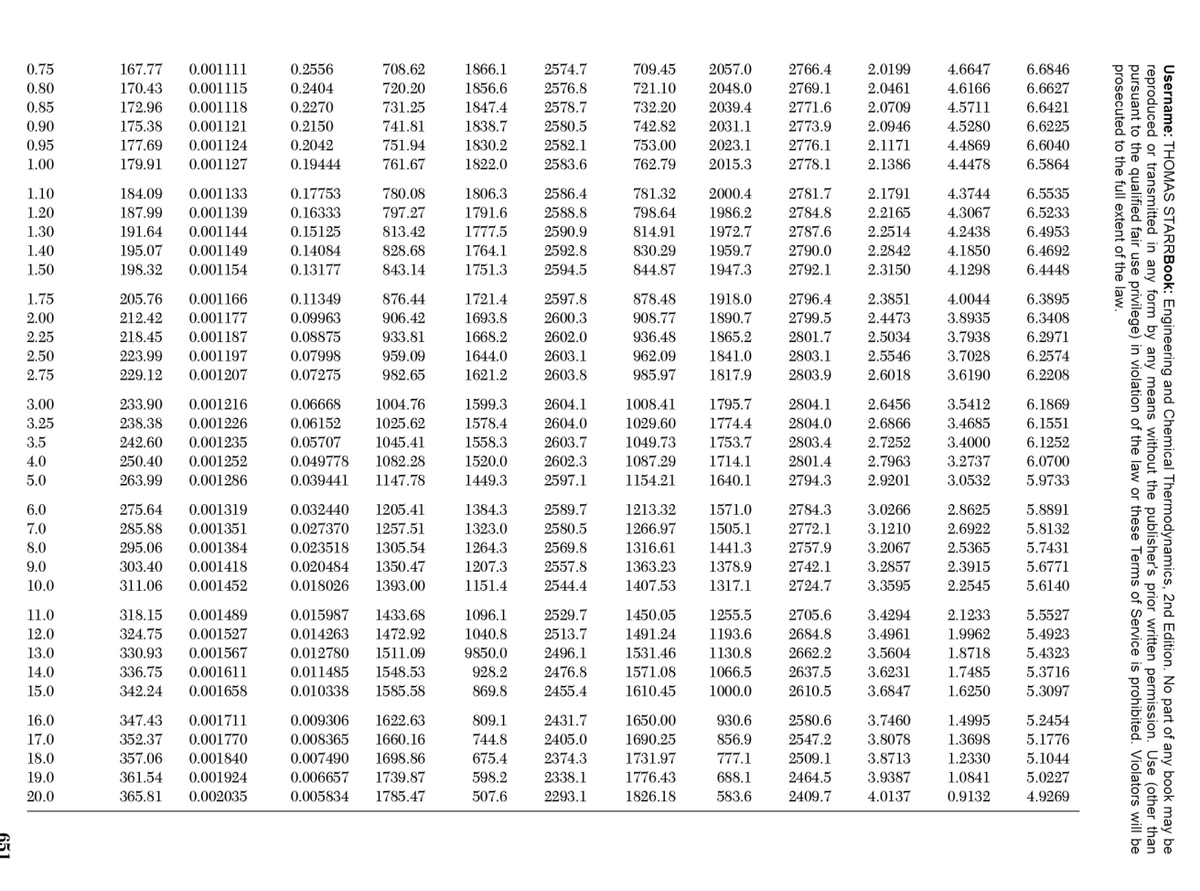
Transcribed Image Text:2766.4
2769.1
0.75
167.77
0.001111
0.2556
708.62
1866.1
2574.7
709.45
2057.0
2.0199
4.6647
6.6846
0.80
170.43
0.001115
0.2404
720.20
1856.6
2576.8
721.10
2048.0
2.0461
4.6166
6.6627
0.001118
0.2270
1847.4
2578.7
2580.5
0.85
172.96
731.25
732.20
2039.4
2771.6
2.0709
4.5711
6.6421
2773.9
2776.1
0.90
175.38
0.001121
0.2150
741.81
1838.7
742.82
2031.1
2.0946
4.5280
6.6225
0.95
177.69
0.001124
0.2042
751.94
1830.2
2582.1
753.00
2023.1
2.1171
4.4869
6.6040
1.00
179.91
0.001127
0.19444
761.67
1822.0
2583.6
762.79
2015.3
2778.1
2.1386
4.4478
6.5864
1.10
184.09
0.001133
0.17753
780.08
1806.3
2586.4
781.32
2000.4
2781.7
2.1791
4.3744
6.5535
1.20
187.99
0.001139
0.16333
797.27
1791.6
2588.8
798.64
1986.2
2784.8
2.2165
4.3067
6.5233
1.30
191.64
0.001144
0.15125
813.42
1777.5
2590.9
814.91
1972.7
2787.6
2.2514
4.2438
6.4953
1.40
195.07
0.001149
0.14084
828.68
1764.1
2592.8
830.29
1959.7
2790.0
2.2842
4.1850
6.4692
1.50
198.32
0.001154
0.13177
843.14
1751.3
2594.5
844.87
1947.3
2792.1
2.3150
4.1298
6.4448
1721.4
1693.8
1.75
205.76
0.001166
0.11349
876.44
2597.8
878.48
1918.0
2796.4
2.3851
4.0044
6.3895
2.00
212.42
0.001177
0.09963
906.42
2600.3
908.77
1890.7
2799.5
2.4473
3.8935
6.3408
2.25
218.45
0.001187
0.08875
933.81
1668.2
2602.0
936.48
1865.2
2801.7
2.5034
3.7938
6.2971
0.07998
2603.1
1841.0
1817.9
2.50
223.99
0.001197
959.09
1644.0
962.09
2803.1
2.5546
3.7028
6.2574
2.75
229.12
0.001207
0.07275
982.65
1621.2
2603.8
985.97
2803.9
2.6018
3.6190
6.2208
3.00
233.90
0.001216
0.06668
1004.76
1599.3
2604.1
1008.41
1795.7
2804.1
2.6456
3.5412
6.1869
3.25
238.38
0.001226
0.06152
1025.62
1578.4
2604.0
1029.60
1774.4
2804.0
2.6866
3.4685
6.1551
3.5
242.60
0.001235
0.05707
1045.41
1558.3
2603.7
1049.73
1753.7
2803.4
2.7252
3.4000
6.1252
3.2737
3.0532
4.0
250.40
0.001252
0.049778
1082.28
1520.0
2602.3
1087.29
1714.1
2801.4
2.7963
6.0700
5.0
263.99
0.001286
0.039441
1147.78
1449.3
2597.1
1154.21
1640.1
2794.3
2.9201
5.9733
6.0
275.64
0.001319
0.032440
1205.41
1384.3
2589.7
1213.32
1571.0
2784.3
3.0266
2.8625
5.8891
7.0
285.88
0.001351
0.027370
1257.51
1323.0
2580.5
1266.97
1505.1
2772.1
3.1210
2.6922
5.8132
8.0
295.06
0.001384
0.023518
1305.54
1264.3
2569.8
1316.61
1441.3
2757.9
3.2067
2.5365
5.7431
9.0
303.40
0.001418
0.020484
1350.47
1207.3
2557.8
1363.23
1378.9
2742.1
3.2857
2.3915
5.6771
10.0
311.06
0.001452
0.018026
1393.00
1151.4
2544.4
1407.53
1317.1
2724.7
3.3595
2.2545
5.6140
11.0
318.15
0.001489
0.015987
1433.68
1096.1
2529.7
1450.05
1255.5
2705.6
3.4294
2.1233
5.5527
12.0
324.75
0.001527
0.014263
1472.92
1040.8
2513.7
1491.24
1193.6
2684.8
3.4961
1.9962
5.4923
330.93
0.012780
2496.1
2662.2
3.5604
5.4323
1.8718
1.7485
13.0
0.001567
1511.09
9850.0
1531.46
1130.8
336.75
1571.08
3.6231
0.011485
0.010338
14.0
0.001611
1548.53
928.2
2476.8
1066.5
2637.5
5.3716
15.0
342.24
0.001658
1585.58
869.8
2455.4
1610.45
1000.0
2610.5
3.6847
1.6250
5.3097
16.0
347.43
0.001711
0.009306
1622.63
809.1
2431.7
1650.00
930.6
2580.6
3.7460
1.4995
5.2454
17.0
352.37
0.001770
0.008365
1660.16
744.8
2405.0
1690.25
856.9
2547.2
3.8078
1.3698
5.1776
18.0
357.06
0.001840
0.007490
1698.86
675.4
2374.3
1731.97
777.1
2509.1
3.8713
1.2330
5.1044
19.0
361.54
0.001924
0.006657
1739.87
598.2
2338.1
1776.43
688.1
2464.5
3.9387
1.0841
5.0227
20.0
365.81
0.002035
0.005834
1785.47
507.6
2293.1
1826.18
583.6
2409.7
4.0137
0.9132
4.9269
Username: THOMAS STARRBook: Engineering and Chemical Thermodynamics, 2nd Edition. No part of any book may be
reproduced or transmitted in any form by any means without the publisher's prior written permission. Use (other than
pursuant to the qualified fair use privilege) in violation of the law or these Terms of Service is prohibited. Violators will be
prosecuted to the full extent of the law.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



