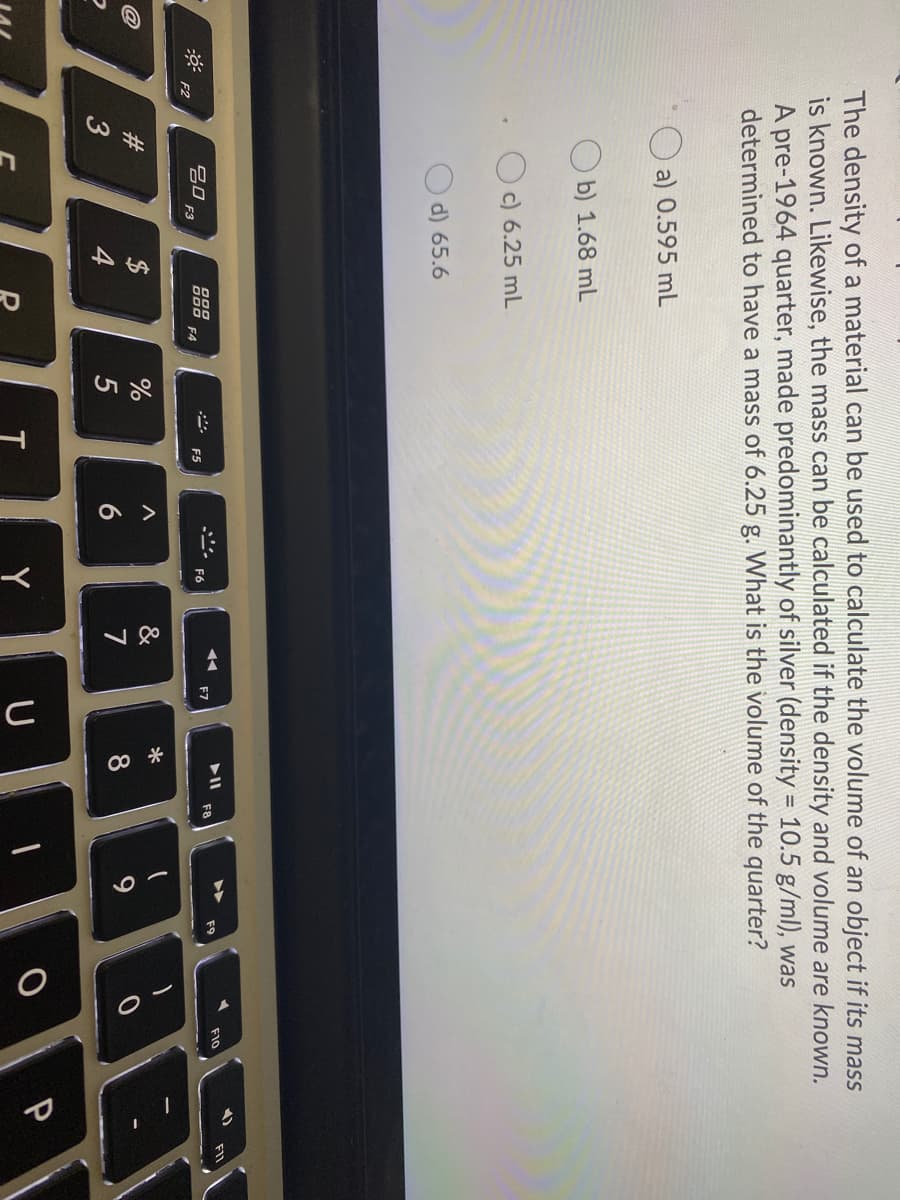
The density of a material can be used to calculate the volume of an object if its mass is known. Likewise, the mass can be calculated if the density and volume are known. A pre-1964 quarter, made predominantly of silver (density = 10.5 g/ml), was determined to have a mass of 6.25 g. What is the volume of the quarter? %3D a) 0.595 mL b) 1.68 mL O c) 6.25 mL d) 65.6
The density of a material can be used to calculate the volume of an object if its mass is known. Likewise, the mass can be calculated if the density and volume are known. A pre-1964 quarter, made predominantly of silver (density = 10.5 g/ml), was determined to have a mass of 6.25 g. What is the volume of the quarter? %3D a) 0.595 mL b) 1.68 mL O c) 6.25 mL d) 65.6
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter3: Matter
Section: Chapter Questions
Problem 9CR
Related questions
Question
Transcribed Image Text:P.
The density of a material can be used to calculate the volume of an object if its mass
is known. Likewise, the mass can be calculated if the density and volume are known.
A pre-1964 quarter, made predominantly of silver (density = 10.5 g/ml), was
determined to have a mass of 6.25 g. What is the volume of the quarter?
a) 0.595 mL
b) 1.68 mL
c) 6.25 mL
d) 65.6
F10
FIL
gO F3
F8
000
F4
F7
F5
F6
F2
&
*
$
4
#3
%
7
8
Y
U
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning