The enamine shown has three significant resonance structures (including the initial structure shown). Modify the giyen drawings as needed to show all three resonance structures. Include formal charges and lone pairs in your answer. Do not drawresonance and curved arrows. * Edit Drawing
The enamine shown has three significant resonance structures (including the initial structure shown). Modify the giyen drawings as needed to show all three resonance structures. Include formal charges and lone pairs in your answer. Do not drawresonance and curved arrows. * Edit Drawing
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter5: Resonance
Section: Chapter Questions
Problem 17CTQ: For each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show...
Related questions
Question
Both of these are part of the same question thank you :)
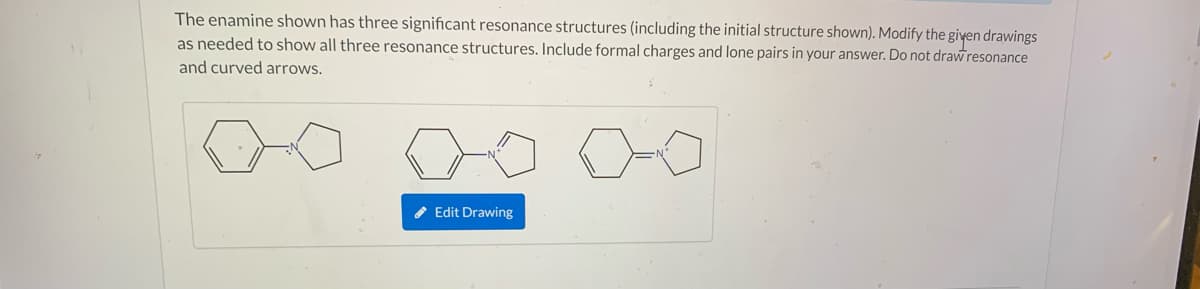
Transcribed Image Text:The enamine shown has three significant resonance structures (including the initial structure shown). Modify the given drawings
as needed to show all three resonance structures. Include formal charges and lone pairs in your answer. Do not drawresonance
and curved arrows.
* Edit Drawing
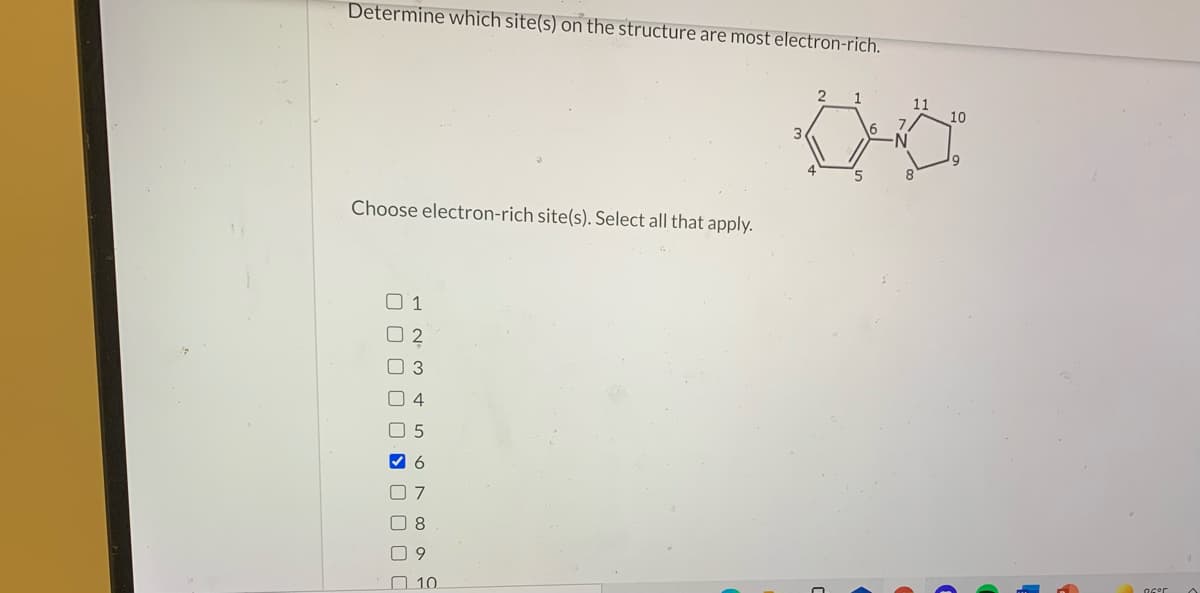
Transcribed Image Text:Determine which site(s) on the structure are most electron-rich.
1.
11
10
3
8.
Choose electron-rich site(s). Select all that apply.
O 3
5
O 8
O 10
O O O O O DO O O C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning