The following energy diagram is the reaction progress for the reaction: (CHg)3CCI + OH" → (CH3)3COH + cr at 45 °C. The energy diagram shows that the reaction mechanism has 2 steps. These 2 steps have the same orientation factors and same number of collisions per second. Which of the following statements concerning the mechanism is correct? 135 kJ- 115 kJ 90 kJ -25 kJ- (CH),CCI + OH reactants -60 kJ + (CH),COH + CF products reaction coordinate (progress of reaction) If a catalyst is added to the above reaction at 45 °C, the rate of the reaction will increase and the heat O a. (enthalpy) of the reaction will decrease for the forward reaction. b. In the mechanism of the above reaction, the activation energy of the first step is 135 k). O c.In the mechanism of the above reaction, the first step is most likely the rate determining step. d. The overall reaction of this reaction is endothermic. e. When the temperature is increased, the activation energy of both steps will decrease. Energy
The following energy diagram is the reaction progress for the reaction: (CHg)3CCI + OH" → (CH3)3COH + cr at 45 °C. The energy diagram shows that the reaction mechanism has 2 steps. These 2 steps have the same orientation factors and same number of collisions per second. Which of the following statements concerning the mechanism is correct? 135 kJ- 115 kJ 90 kJ -25 kJ- (CH),CCI + OH reactants -60 kJ + (CH),COH + CF products reaction coordinate (progress of reaction) If a catalyst is added to the above reaction at 45 °C, the rate of the reaction will increase and the heat O a. (enthalpy) of the reaction will decrease for the forward reaction. b. In the mechanism of the above reaction, the activation energy of the first step is 135 k). O c.In the mechanism of the above reaction, the first step is most likely the rate determining step. d. The overall reaction of this reaction is endothermic. e. When the temperature is increased, the activation energy of both steps will decrease. Energy
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.96PAE: The following statements relate to the reaction for the formation of HI: H2(g) + I2(g) -* 2 HI(g)...
Related questions
Question
please help
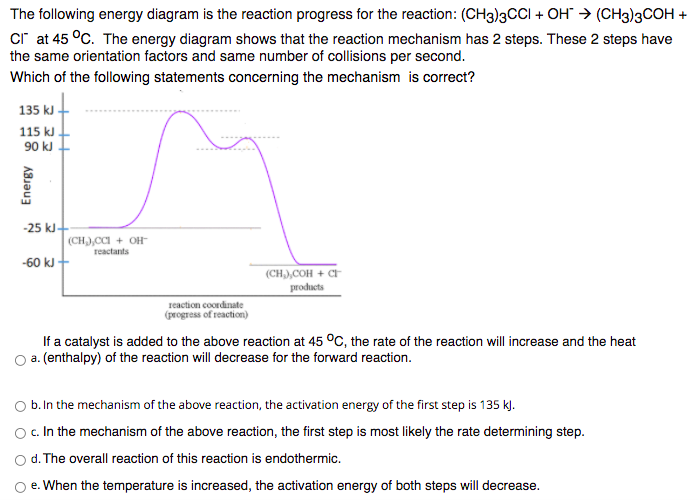
Transcribed Image Text:The following energy diagram is the reaction progress for the reaction: (CH3)3CCI + OH" → (CH3)3COH +
cr at 45 °C. The energy diagram shows that the reaction mechanism has 2 steps. These 2 steps have
the same orientation factors and same number of collisions per second.
Which of the following statements concerning the mechanism is correct?
135 kJ
115 kJ
90 kJ
-25 kJ-
(CH),CCI + OH-
reactants
-60 kJ
(CH),COH + CH
products
reaction coordinate
(progress of reaction)
If a catalyst is added to the above reaction at 45 °C, the rate of the reaction will increase and the heat
a. (enthalpy) of the reaction will decrease for the forward reaction.
b. In the mechanism of the above reaction, the activation energy of the first step is 135 k).
O c.In the mechanism of the above reaction, the first step is most likely the rate determining step.
d. The overall reaction of this reaction is endothermic.
e. When the temperature is increased, the activation energy of both steps will decrease.
Energy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning