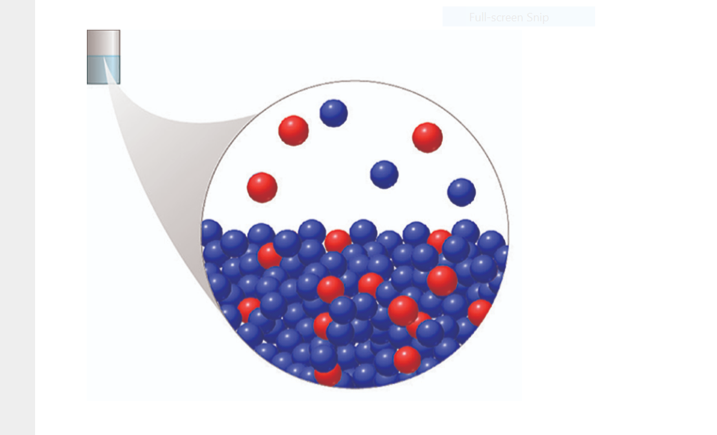
The following figures shows a particle-level view of a sealed container partially filled with a solution of two miscible liquids: X (blue spheres) and Y (red spheres). Which of the following statements about substances X and Y are true? Assume ideal behavior. Choose one or more: A.The presence of Y in the solution lowers the vapor pressure of X. B.If Y were not present, there would be more X particles in the gas above the liquid solution. C.Y is the solvent in this solution. D.Pure Y has a higher vapor pressure than pure X.
The following figures shows a particle-level view of a sealed container partially filled with a solution of two miscible liquids: X (blue spheres) and Y (red spheres). Which of the following statements about substances X and Y are true? Assume ideal behavior. Choose one or more: A.The presence of Y in the solution lowers the vapor pressure of X. B.If Y were not present, there would be more X particles in the gas above the liquid solution. C.Y is the solvent in this solution. D.Pure Y has a higher vapor pressure than pure X.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter1: Introduction
Section: Chapter Questions
Problem 1.7QAP
Related questions
Question
The following figures shows a particle-level view of a sealed container partially filled with a solution of two miscible liquids: X (blue spheres) and Y (red spheres).
Which of the following statements about substances X and Y are true? Assume ideal behavior.
Choose one or more:
A.The presence of Y in the solution lowers the vapor pressure of X.
B.If Y were not present, there would be more X particles in the gas above the liquid solution.
C.Y is the solvent in this solution.
D.Pure Y has a higher vapor pressure than pure X.
Transcribed Image Text:Full-screen Snip
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER