The following mixtures are completely mixed to form mixture D: Mixture Mass (kg) % NaCI % KCI % H2O A 50 50 50 В 100 20 80 C 75 40 60 1. Adding first mixtures A and B to yield the resultant mixture F. Then, add mixture F to mixture C to locate mixture D. 2. If 25 kg of H20 was removed from mixture D to produce mixture E, locate E and determine its composition.
The following mixtures are completely mixed to form mixture D: Mixture Mass (kg) % NaCI % KCI % H2O A 50 50 50 В 100 20 80 C 75 40 60 1. Adding first mixtures A and B to yield the resultant mixture F. Then, add mixture F to mixture C to locate mixture D. 2. If 25 kg of H20 was removed from mixture D to produce mixture E, locate E and determine its composition.
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter3: Melting Points And Boiling Points
Section: Chapter Questions
Problem 8Q
Related questions
Question
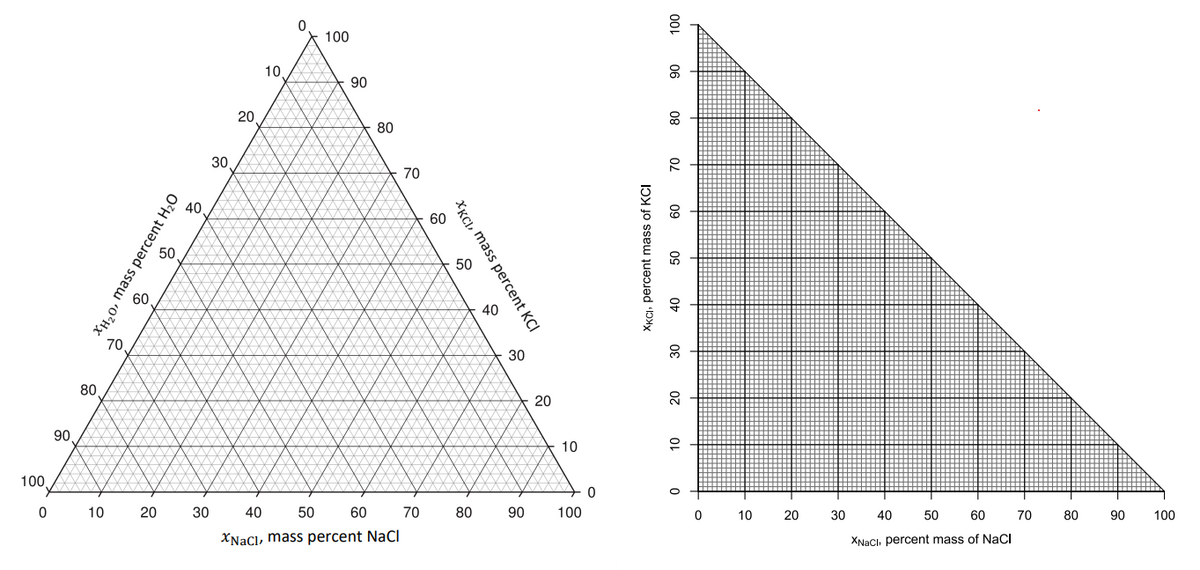
Transcribed Image Text:Using (i) a right triangular plot and (ii) an equilateral triangular plot, solve the following problem graphically.
The following mixtures are completely mixed to form mixture D:
Mixture
Mass (kg)
% NaCI
% KCI
% H2O
A
50
50
50
В
100
20
80
C
75
40
60
1. Adding first mixtures A and B to yield the resultant mixture F. Then, add mixture F to mixture C to locate mixture
D.
2. If 25 kg of H2O was removed from mixture D to produce mixture E, locate E and determine its composition.
Transcribed Image Text:100
10.
90
20
80
30,
70
40
60
So
60
40
40
70
30
30
80
20
20
90,
10
100
10
20
30
40
50
60
70
80
90
100
XNaCl, mass percent NaCl
10
20
30
40 50
60
70
80
90
100
XNaCl. percent mass of NaCI
06
09
09
XKCI, percent mass of KCI
XKCI, mass percent KCI
XH,0, mass percent H20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole