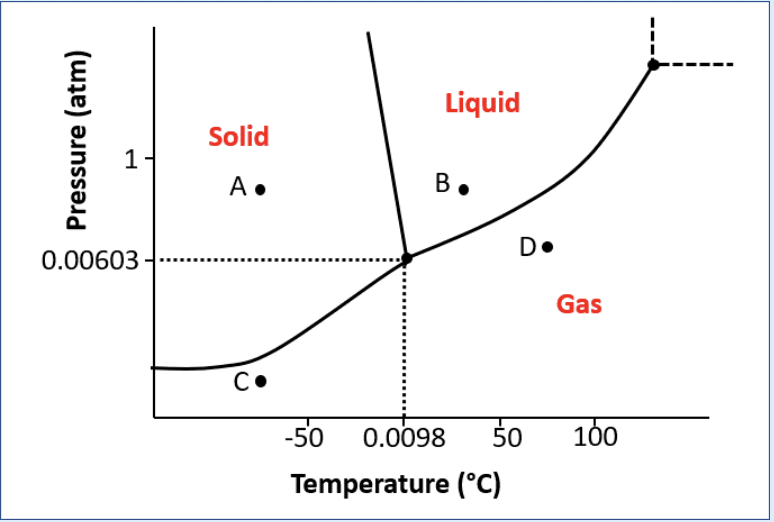
The following phase diagram for water was given: a) Complete the following sentences regarding navigation from one point to another across the phase diagram given. answer box: only temperature increases only the pressure increases the temperature and pressure increase only the temperature decreases only the temperature increases the temperature and pressure decrease the temperature increases and the pressure decreases the pressure increases and the temperature decreases 1. Moving from point A to B 2. Moving from point C to D 3. Moving from point D to B 4. Moving from point A to C
The following phase diagram for water was given: a) Complete the following sentences regarding navigation from one point to another across the phase diagram given. answer box: only temperature increases only the pressure increases the temperature and pressure increase only the temperature decreases only the temperature increases the temperature and pressure decrease the temperature increases and the pressure decreases the pressure increases and the temperature decreases 1. Moving from point A to B 2. Moving from point C to D 3. Moving from point D to B 4. Moving from point A to C
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.77PAE
Related questions
Question
The following phase diagram for water was given:
a) Complete the following sentences regarding navigation from one point to another across the phase diagram given.
answer box:
| only temperature increases | only the pressure increases | the temperature and pressure increase | only the temperature decreases |
| only the temperature increases | the temperature and pressure decrease | the temperature increases and the pressure decreases | the pressure increases and the temperature decreases |
1. Moving from point A to B
2. Moving from point C to D
3. Moving from point D to B
4. Moving from point A to C
Transcribed Image Text:Pressure (atm)
1
0.00603
Solid
A.
C•
Liquid
B.
D.
Gas
-50 0.0098 50 100
Temperature (°C)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning