The following table shows the distance moved in an SDS-polyacrylamide gel by a series of marker proteins of known relative molecular mass ( M, ). A newly purified protein (X) run on the same gel showed a single band that had moved a distance of 55 mm. What is the M, of protein X if the tracking dye covered a distance of 95 mm? Round off your answer to the nearest whole number. Protein M. Distance moved (mm) Transferrin 78 000 6.0 Bovine serum albumin 66 000 12.5 Ovalbumin 45 000 32.0 38.0 Glyceraldehyde-3- phosphate dehydrogenase carbonic anhydrase 36 000 29 000 50.0 Trypsinogen 24 000 54.0 Soyabean trypsin inhibitor 20 100 61.0 Myoglobin 17 800 69.0 Lysozyme 14 300 79.0 Cytochrome c 12 400 86.5
The following table shows the distance moved in an SDS-polyacrylamide gel by a series of marker proteins of known relative molecular mass ( M, ). A newly purified protein (X) run on the same gel showed a single band that had moved a distance of 55 mm. What is the M, of protein X if the tracking dye covered a distance of 95 mm? Round off your answer to the nearest whole number. Protein M. Distance moved (mm) Transferrin 78 000 6.0 Bovine serum albumin 66 000 12.5 Ovalbumin 45 000 32.0 38.0 Glyceraldehyde-3- phosphate dehydrogenase carbonic anhydrase 36 000 29 000 50.0 Trypsinogen 24 000 54.0 Soyabean trypsin inhibitor 20 100 61.0 Myoglobin 17 800 69.0 Lysozyme 14 300 79.0 Cytochrome c 12 400 86.5
Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.2QAP
Related questions
Question
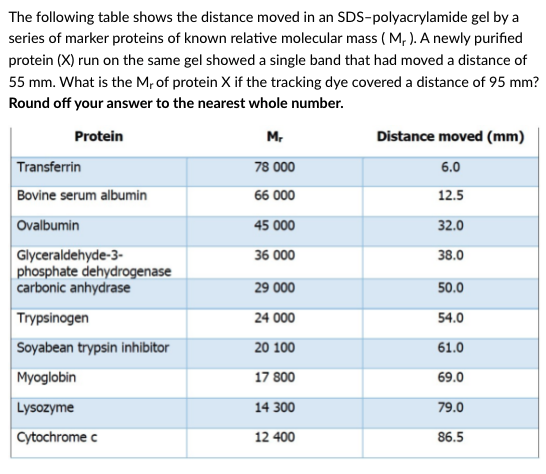
Transcribed Image Text:The following table shows the distance moved in an SDS-polyacrylamide gel by a
series of marker proteins of known relative molecular mass ( M, ). A newly purified
protein (X) run on the same gel showed a single band that had moved a distance of
55 mm. What is the M, of protein X if the tracking dye covered a distance of 95 mm?
Round off your answer to the nearest whole number.
Protein
M.
Distance moved (mm)
Transferrin
78 000
6.0
Bovine serum albumin
66 000
12.5
Ovalbumin
45 000
32.0
Glyceraldehyde-3-
phosphate dehydrogenase
carbonic anhydrase
36 000
38.0
29 000
50.0
Trypsinogen
24 000
54.0
Soyabean trypsin inhibitor
20 100
61.0
Myoglobin
17 800
69.0
Lysozyme
14 300
79.0
Cytochrome c
12 400
86.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning