The graph shows the reaction pathways for the same reaction: One reaction pathway shows an uncatalyzed reaction, while the other reaction pathway shows a catalyzed reaction. Label the graph. Drag each label to the appropriate target. > View Available Hint(s) Reset Help A Transition state uncatalyzed reaction activation energy of uncatalyzed reaction В В activation energy of catalyzed reaction catalyzed reaction Reaction pathway Energy
The graph shows the reaction pathways for the same reaction: One reaction pathway shows an uncatalyzed reaction, while the other reaction pathway shows a catalyzed reaction. Label the graph. Drag each label to the appropriate target. > View Available Hint(s) Reset Help A Transition state uncatalyzed reaction activation energy of uncatalyzed reaction В В activation energy of catalyzed reaction catalyzed reaction Reaction pathway Energy
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 71QAP: For a certain reaction, Ea is 135 kJ and H=45 kJ. In the presence of a catalyst, the activation...
Related questions
Question
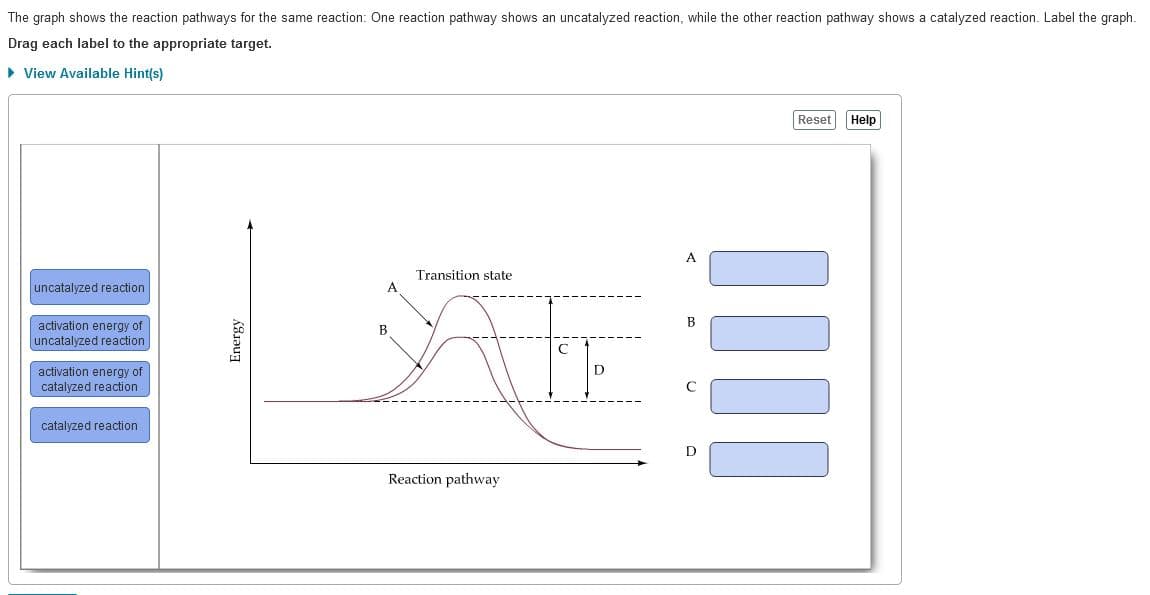
Transcribed Image Text:The graph shows the reaction pathways for the same reaction: One reaction pathway shows an uncatalyzed reaction, while the other reaction pathway shows a catalyzed reaction. Label the graph.
Drag each label to the appropriate target.
• View Available Hint(s)
Reset
Help
A
Transition state
uncatalyzed reaction
B
activation energy of
uncatalyzed reaction
B
activation energy of
catalyzed reaction
catalyzed reaction
Reaction pathway
Energy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning