The heat from a candy bar's Calories can raise the temperature of a 4.50 kg bucket of water from 4°C to 68°C. How many Calories are in the candy bar?
The heat from a candy bar's Calories can raise the temperature of a 4.50 kg bucket of water from 4°C to 68°C. How many Calories are in the candy bar?
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter2: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 2.14E: A 5-mmdiameter hailstone has a terminal velocity of10.0m/s. Assuming its mass is6.031025 kg and all...
Related questions
Question
5,6,7
Transcribed Image Text:wwwntro N
Nearpod W
Classwork fe
docs google.com/document/d/1vJOAin W50HetaGTXANTIlDEKSNuYOGI6C7 P3Mo/edit
New folder
Genetic drift Wikip
4 Algebra Foundation.
Image result
Surrealam-The M.
Morris-Specific Heat Practice @
View Insert Format Tools Add ons Help Accessibility
Last edit
- - II B IUA
100%
Normal text
Arial
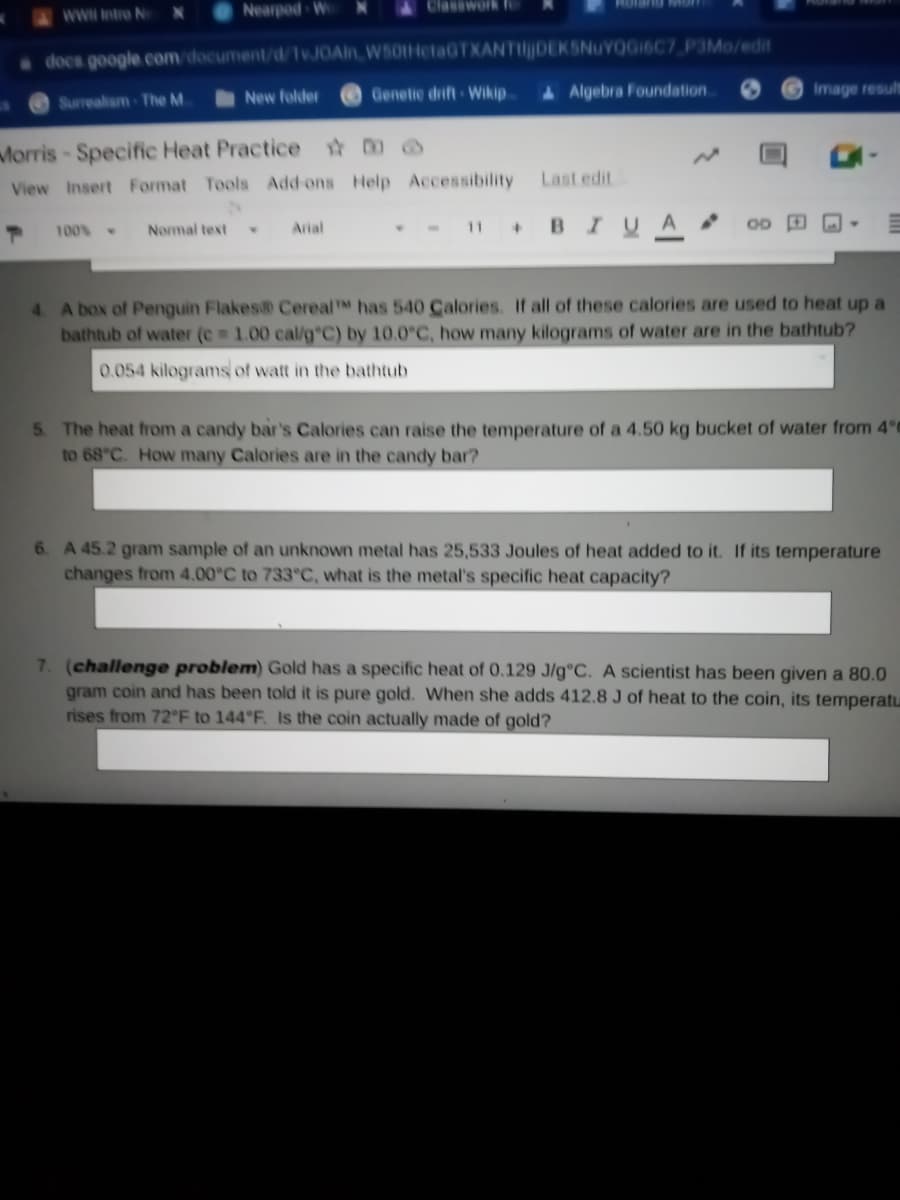
4 A box of Penguin Flakes CerealTM has 540 Calories. If all of these calories are used to heat up a
bathtub of water (e 1.00 cal/g C) by 10.0°C, how many kilograms of water are in the bathtub?
0.054 kilograms of watt in the bathtub
5 The heat from a candy bar's Calories can raise the temperature of a 4.50 kg bucket of water from 4°C
to 68°C. How many Calories are in the candy bar?
6. A 45.2 gram sample of an unknown metal has 25,533 Joules of heat added to it. If its temperature
changes from 4.00°C to 733°C, what is the metal's specific heat capacity?
7. (challenge problem) Gold has a specific heat of 0.129 J/g°C. A scientist has been given a 80.0
gram coin and has been told it is pure gold. When she adds 412.8 J of heat to the coin, its temperatu
rises from 72°F to 144°F. Is the coin actually made of gold?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,