The heating curve shown was generated by measuring the heat flow and temperature for a sample of ice as it was heated. The heat flow into the sample in the segment will yield the value of the AHvap of this sample. Heating curve for 1.00 mol of ice at -25°C 125 F Water vapor D 100 Liquid water and vapor (vaporization) 75 50 Liquid water 25 в с Ice and liquid water (melting) Ice A -25 Heat added (each division corresponds to 4 kJ) Select one: O a. DE O b. AB О с. ВС O d. CD Temperature (°C)
The heating curve shown was generated by measuring the heat flow and temperature for a sample of ice as it was heated. The heat flow into the sample in the segment will yield the value of the AHvap of this sample. Heating curve for 1.00 mol of ice at -25°C 125 F Water vapor D 100 Liquid water and vapor (vaporization) 75 50 Liquid water 25 в с Ice and liquid water (melting) Ice A -25 Heat added (each division corresponds to 4 kJ) Select one: O a. DE O b. AB О с. ВС O d. CD Temperature (°C)
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter15: Gases,liquids, And Solids
Section: Chapter Questions
Problem 97E
Related questions
Question
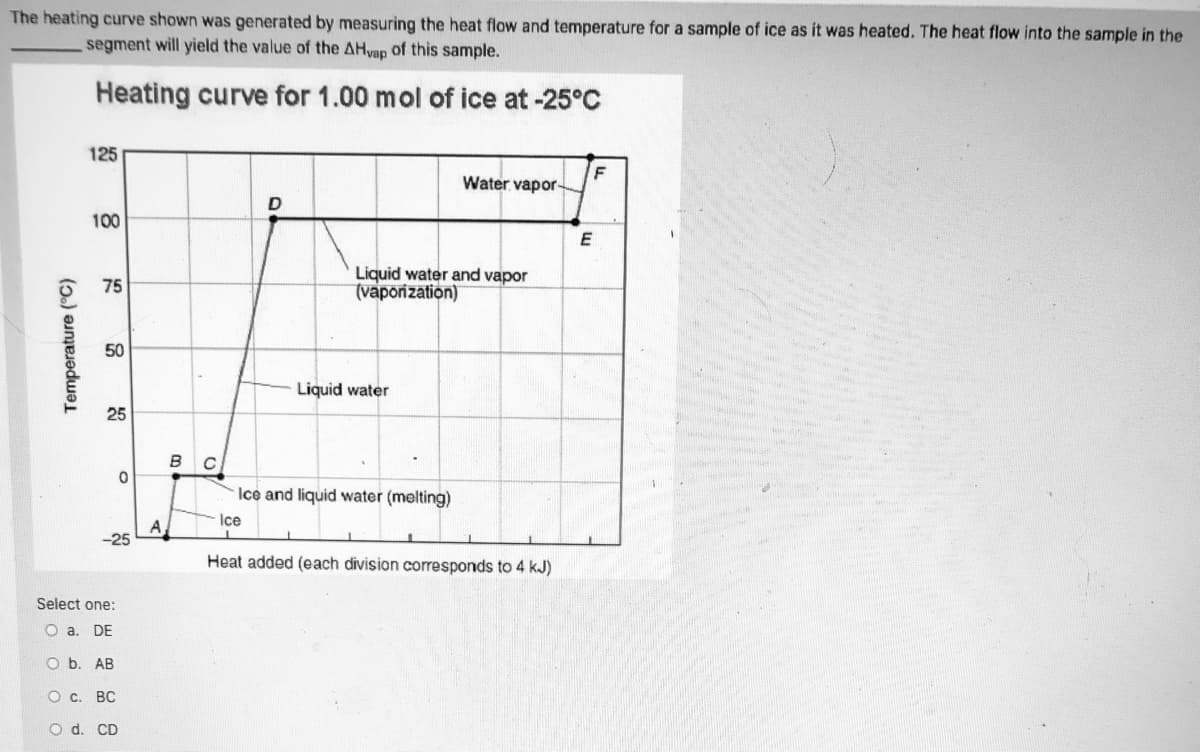
Transcribed Image Text:The heating curve shown was generated by measuring the heat flow and temperature for a sample of ice as it was heated. The heat flow into the sample in the
segment will yield the value of the AHvap of this sample.
Heating curve for 1.00 mol of ice at -25°C
125
Water vapor-
D
100
Liquid water and vapor
(vaporization)
75
50
Liquid water
25
в с
Ice and liquid water (melting)
Ice
A
-25
Heat added (each division corresponds to 4 kJ)
Select one:
O a. DE
O b. AB
О с. ВС
O d. CD
Temperature (°C)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning