The high-pressure phase diagram of ice is shown here. Notice that, under high pressure, ice can exist in several different solid forms. What three forms of ice are present at the triple point marked O? How does the density of ice II compare to ice I (the familiar form of ice)? Would ice III sink or float in liquid water? Ice VII Ice VI Ice V Ice II Ice I 1 atm Ice l Liquid water Gaseous water Temperature (not to scale) Pressure (not to scale)
The high-pressure phase diagram of ice is shown here. Notice that, under high pressure, ice can exist in several different solid forms. What three forms of ice are present at the triple point marked O? How does the density of ice II compare to ice I (the familiar form of ice)? Would ice III sink or float in liquid water? Ice VII Ice VI Ice V Ice II Ice I 1 atm Ice l Liquid water Gaseous water Temperature (not to scale) Pressure (not to scale)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter9: Liquids And Solids
Section: Chapter Questions
Problem 11QAP: At a resort in Sante Fe, New Mexico, the barometric pressure is 625 mm Hg. Water boils in an open...
Related questions
Question
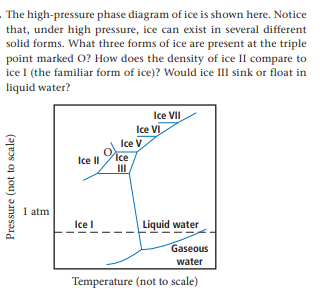
Transcribed Image Text:The high-pressure phase diagram of ice is shown here. Notice
that, under high pressure, ice can exist in several different
solid forms. What three forms of ice are present at the triple
point marked O? How does the density of ice II compare to
ice I (the familiar form of ice)? Would ice III sink or float in
liquid water?
Ice VII
Ice VI
Ice V
Ice
II
Ice I
1 atm
Ice l
Liquid water
Gaseous
water
Temperature (not to scale)
Pressure (not to scale)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning