The image below shows the Lineweaver-Burke plot for Enzyme catalysed reaction. Units for the Y- axis is (s/mmol) and X-axis is (1/mM). Use the data on the plot to calculate Vmax and Km for this reaction. Type your answers in the text box below clearly indicating which value is Vmax and which one is Km. You do not need to show your workings but don't forget your units. -0.4 Download -0.2 0.4 0.3 0.2 0.1 ..0.0. -0.1 0.0 ENZYME CATALYSED REACTION 0.2 0.4 y = 0.3086x + 0.0276 R² = 0.9967 0.6 0.8 1.0 1.2
The image below shows the Lineweaver-Burke plot for Enzyme catalysed reaction. Units for the Y- axis is (s/mmol) and X-axis is (1/mM). Use the data on the plot to calculate Vmax and Km for this reaction. Type your answers in the text box below clearly indicating which value is Vmax and which one is Km. You do not need to show your workings but don't forget your units. -0.4 Download -0.2 0.4 0.3 0.2 0.1 ..0.0. -0.1 0.0 ENZYME CATALYSED REACTION 0.2 0.4 y = 0.3086x + 0.0276 R² = 0.9967 0.6 0.8 1.0 1.2
Chapter13: Kinetic Methods
Section: Chapter Questions
Problem 4P
Related questions
Question
Kk161.
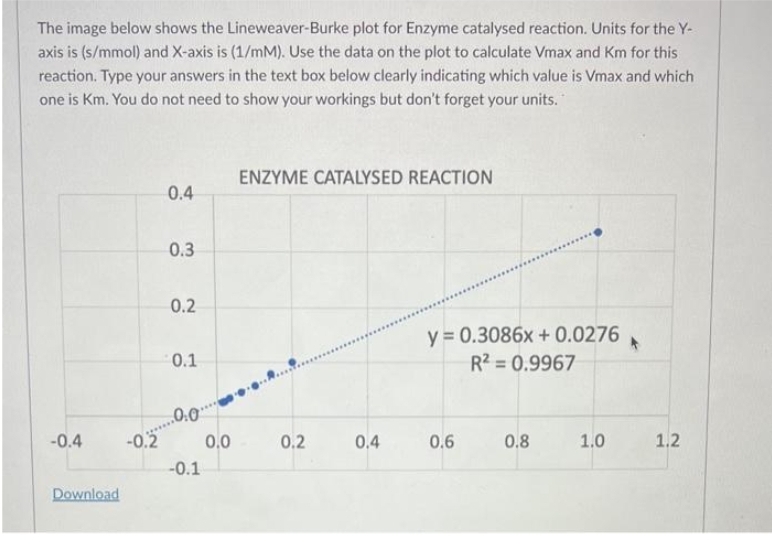
Transcribed Image Text:The image below shows the Lineweaver-Burke plot for Enzyme catalysed reaction. Units for the Y-
axis is (s/mmol) and X-axis is (1/mM). Use the data on the plot to calculate Vmax and Km for this
reaction. Type your answers in the text box below clearly indicating which value is Vmax and which
one is Km. You do not need to show your workings but don't forget your units.
-0.4
Download
-0.2
0.4
0.3
0.2
0.1
..0.0
-0.1
0.0
ENZYME CATALYSED REACTION
0.2
0.4
y = 0.3086x + 0.0276 *
R² = 0.9967
0.6
0.8
1.0
1.2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

