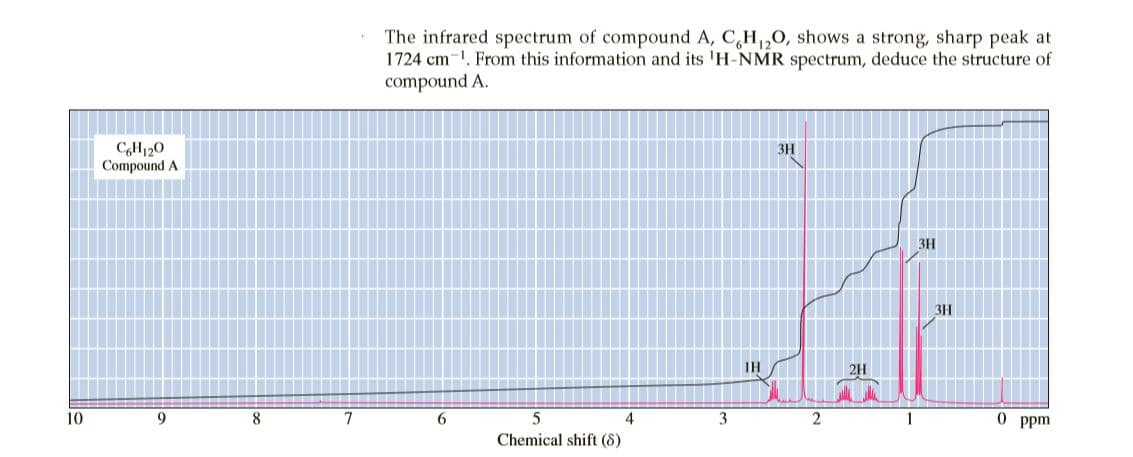
The infrared spectrum of compound A, C,H,0, shows a strong, sharp peak at 1724 cm-1. From this information and its 'H-NMR spectrum, deduce the structure of compound A. CH120 Compound A ЗН 3H 3H IH 2H 10 9. 8. 7 6. 4 3 1 O ppm Chemical shift (8)
Q: An aromatic compound, C9H12 has the following 1H NMR spectrum and a peak at 750 cm-1 in its IR…
A: Aromatic compound represents the presence of benzene ring. A characteristic IR peak at 750 cm-1…
Q: The structure of an ester could either be A or B: CH3 O O CH3 T || -C-OCH3 CH3-C CH3-C-0-C-CH3 CH3…
A: An instrumentational method that is used to determine the structure of a compound by analyzing its…
Q: Write the equation for the reaction of the following with acetaldehyde: a. 2,4-DNP b. Schiff's…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: A compound of molecular formula C5H12O2 compound has strong infrared absorption at 3300 to 3400…
A: 1. Degree of unsaturation (D.O.U) : D.O.U = (2C + 2 - H + N - X ) / 2 = ( 2 x 5 + 2 -…
Q: An aromatic compound C8H9Br has the following 1H NMR spectrum and a peak at 820 cm-1 in its IR…
A:
Q: 9a) The proton bonded to the carbon adjacent to the carbonyl group in the following compound can be…
A: Chiral Centre: atom which is bonded with four different atoms /groups is called chiral Centre
Q: Which of the following reaction sequences would convert 2- butanol into the deuterated compound…
A: The correct reaction sequence would convert 2-butanol into the deuterated compound can be shown…
Q: In the IR spectrum, a compound shows a strong absorption in the region between 3300-3600 cm and…
A:
Q: What are the infrared absorption frequency characteristics that would distinguish the following…
A: The inspecting of an unknown compound by its molecular formula is done by calculating the index of…
Q: Which set of reagents would be appropriate to synthesize bromobenzene from benzene? O1. HNO3 in…
A: HNO3 in H2SO4 gives nitration to benzene ring. Here, electrophilic substitution reaction takes…
Q: Sketch and explain the IR, UV, mass, and NMR (1H and 13C) spectroscopy data of Benzonitrile
A: There are different types of spectroscopic techniques for the identification and characterisation of…
Q: Identify products A and B from the given 1H NMR data. Treatment of CH2=CHCOCH3 with one equivalent…
A: Addition of a molecule over a double bond is called addition reaction. After addition of HCl, three…
Q: The 1H and 13C NMR spectra of compound A, C8H9Br are shown below. Answer the following questions.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Suppose you have compounds A-D at your disposal. Using these compounds, devise two different ways to…
A: (i) A + D -------> E ( major product) (ii) B + C -------> E ( minor product) Method (i) is…
Q: Give the structural formulas of compounds A, B, and C in the following problems. EXPLAIN EXACTLY HOW…
A: 2,4-dinitrophenylhydrazine test is mainly used for the identification of the presence of aldehyde or…
Q: Benzene magnesium bromide (Grignard reagent) reacts with acetaldehyde in diethyl ether to produce a…
A:
Q: IH NMR spectrum of methyl-m-nitrobenzoate 'H nmr, 8: 8.86 (t, IH), 842(dd, IH), 8.37 (dd, IH), 7.67…
A: There are two types of equivalency-Chemical and magnetic. Chemically non-equivalent hydrogens show…
Q: A retrosynthetic analysis of 1 was undertaken suggesting a forward synthesis starting from compounds…
A: Epoxides can be converted to aminoalcohols by the reaction of epoxide directly with ammonia or first…
Q: 8 Below is the H-NMR of a reaction product of aniline. Provide the structure of the product and the…
A:
Q: b) A compound with molecular formula C,H1402 upon hydrolysis produces an alcohol and an acid. It has…
A:
Q: 8. (a) Benzene derivatives exhibit medium to strong absorption in UV-region. Explain why aniline and…
A: Firstly we need to know about UV region abosorbtion. Each type of spectroscopy works differently. UV…
Q: A compound containing one functional group has IR absorption bands at 3300 cm1 (strong, sharp) and…
A:
Q: Which structure corresponds to the ester compound being referred to based on the following chemical…
A:
Q: When acetonitrile is treated with concentrated sulfuric acid and tert-butanol, followed by water, a…
A: The given reaction is,
Q: Primary amines can also be prepared by the reaction of an alkyl halide with azide ion, followed by…
A:
Q: An unknown compound A (molecular formula C7H14O) was treated withNaBH4 in CH3OH to form compound B…
A: The nuclear magnetic resonance spectroscopy or NMR deals with the study of magnetic properties of…
Q: A compound shows only one peak at 0.9 ppm on its 1H-NMR. Its IR shows only a strong peak right…
A: One peak at 0.9 ppm on its 1H-NMR indicates the presence of only aliphatic protons. So the molecule…
Q: Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that…
A: The 1H NMR peak between 7-8 ppm signifies the presence of benzene ring. Triplet at approximately…
Q: Friedel- Crafts Acylation correlate the boiling points of toluene and 1-(p-tolyl) ethanone with…
A: Friedel- Crafts Acylation: Electrophilic aromatic substitution reactions occur between the…
Q: What is the structure of the compound with the formula C4H7ClO, it has a strong IR signal near 1730…
A:
Q: An unknown compound A (molecular formula C7H14O) was treated with NaBH4 in CH3OH to form compound B…
A: The molecular formula of the compound A is given C7H14O Compound A has strong absorption in its IR…
Q: Phenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in…
A: Molecular formula of the compound is C10H13NO2 Since the compound contains Nitrogen atom in the…
Q: A retrosynthetic analysis of 1 was undertaken suggesting a forward synthesis starting from compounds…
A: We have to synthesize organic compound 1 :
Q: Carbon NMR Spectrum A has peaks at delta 71, 40, 19, and 12. Spectrum B has peaks at delta 72, 38,…
A: According to the given data we have Spectrum A and spectrum B whose C NMR values are being given…
Q: A compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes…
A:
Q: When a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the…
A:
Q: An unknown compound A (molecular formula C7H14O) was treated with NaBH4 in CH3OH to form compound B…
A: Calculation of double bond equivalence: DBE = C +1 – (H + X – N) / 2 Here, C is the number of carbon…
Q: . explain each step with words on how to get the structure. Step 1: Calculate the Index of…
A:
Q: An unknown compound C of molecular formula C6H12O3 exhibits a strong absorption in its IR spectrum…
A: The molecular formula of the compound C is C6H12O3. Information from IR data: The IR absorption at…
Q: 8. A compound with a molecular formula C,H1,CIO, has the following 'H NMR spectrum. The IR spectrum…
A: 1H NMR- It is application of NMR spectroscopy called as proton Nuclear Magnetic resonance…
Q: M-C-H AICI3 t. HBr D + [o] room temperature (A)
A:
Q: Sketch and explain the IR, UV, mass, and NMR (1H and 13C) spectroscopy data of benzaldehyde.
A: In IR spectroscopy, characteristic infrared bands in the range 2,890-2,660 for C-H aldehyde, 3073…
Q: Compound A is treated with a mixture of nitric and sulfuric acids to generate Compound B. The 1H-NMR…
A:
Q: draw the strucutre based off the IR spectrum, 1H NMR spectrum, and 13C NMR spectrum for this…
A:
Q: What peaks are expected in the IR spectrum and NMR (C-NMR and P-NMR) spectrum of triphenyl triethyl…
A: Here we consider the decoupled NMR , where peaks do not further splitted .
Q: Show the structures of the missing substance(s) in each of the following acid-base equilibria. a.…
A: Nitrogen is lewis base which can accept hydrogen from acid and form positive charge on it.
Q: Indene NH4Br, KHSO5, CH3CN:H₂O Br OH or "Bromoindanol" OH Br You will determine the structure of…
A: This occur via bromonium intermidiate
Q: As a method for the synthesis of cinnamaldehyde (3-phenyl-2-propenal), a chemist treated…
A:
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

- Compound A with molecular formula C6H10 has two peaks in its 1H NMR spectrum, both of which are singlets (with ratio 9 : 1). Compound A reacts with an acidic aqueous solution containing mercuric sulfate to form compound B, which gives a positive iodoform test and has an 1H NMR spectrum that shows two singlets (with ratio 3 : 1). Identify A and B.There is a Compound called A with the formula C13H20N2O2. In the proton NMR two triplets at 2.8 and 4.3 are coupled to each other. Similarly, a triplet at 1.1 and a quartet at 2.6 are coupled to each other. the carbonyl group appearing at 1669 cm -1 in the IR spectrum has an unusually low value. Please provide the structure of A.An unknown compound A (molecular formula C7H14O) was treated withNaBH4 in CH3OH to form compound B (molecular formula C7H16O).Compound A has a strong absorption in its IR spectrum at 1716 cm−1.Compound B has a strong absorption in its IR spectrum at 3600−3200cm−1. The 1H NMR spectra of A and B are given. What are the structuresof A and B?
- Compounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.Treatment of isobutene [(CH3)2C = CH2] with (CH3)3CLi forms a carbanion that reacts with CH2=O to form H after water is added to the reaction mixture. H has a molecular ion in its mass spectrum at m/z = 86, and shows fragments at 71 and 68. H exhibits absorptions in its IR spectrum at 3600–3200 and 1651 cm−1, and has the 1H NMR spectrum given below. Whatis the structure of H?Reaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G?
- Shown are the H NMR spectra for 2 isomeric compounds of the formula C5H10O. The IR spectrum of both have an absorption in the region of 1700 to 1730 cm-1. Provide the structure for each compound, and which hydrogen atoms give rise to the peaks in each spectrum. The peak at 7.27 ppm can be ignored, and the red numbers are integration values.What is the structure of the compound with the formula C10H10O4, if it shows strong IR signal near 3050 and 1730 cm-1, the 1H-NMR spectrum shows only two signals, both singlets. One of them appears at 3.9 ppm with the relative integration value of 79 and the second one appears at 8.1 ppm with the relative integration of 52.Reaction of C6H5CH2CH2OH with CH3COCl affords compound W, whichhas molecular formula C10H12O2. W shows prominent IR absorptions at3088–2897, 1740, and 1606 cm−1. W exhibits the following signals in its1H NMR spectrum: 2.02 (singlet), 2.91 (triplet), 4.25 (triplet), and 7.20–7.35(multiplet) ppm. What is the structure of W?
- What spectral features including mass spectra, IR spectra, proton spectra and carbon spectra allow you to differentiate the product (Benzonitrile) from the starting material (Benzoic Acid)? Show IR,MS,HNMR,CNMR seperatlyShown are the H NMR spectra for 2 isomeric compounds three and four of the formula C5H10O. The IR spectrum of both have an absorption in the region of 1700 to 1730 cm-1. Provide the structure for each compound, and which hydrogen atoms give rise to the peaks in each spectrum. The peak at 7.27 ppm can be ignored, and the red numbers are integration values.A reaction with compound X (C8H15OCl) with water produces a product which has an absorbance in the IR spectrum at 1800 and 3500 cm-1. What fictional group is present?

