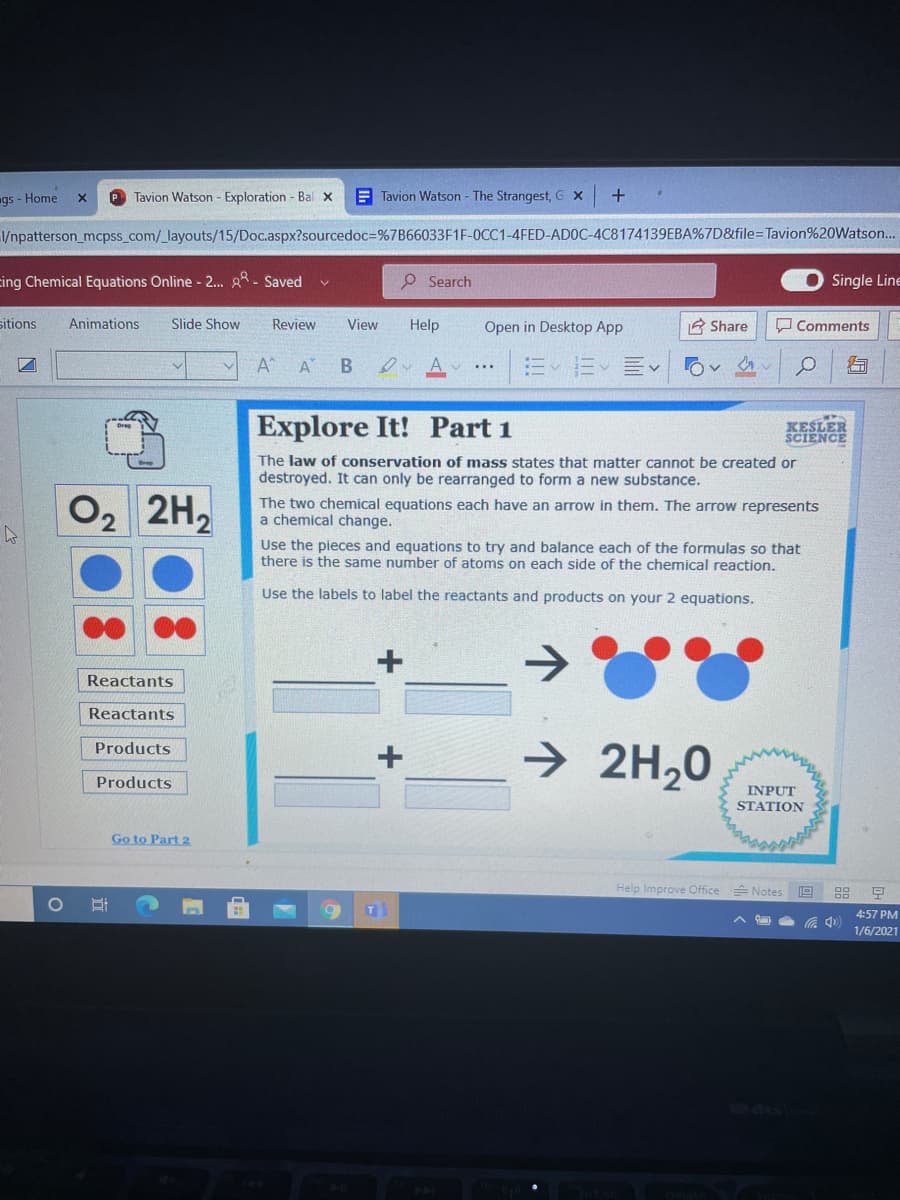
The law of conservation of mass states that matter cannot be created or destroyed. It can only be rearranged to form a new substance. The two chemical equations each have an arrow in them. The arrow represents a chemical change. Use the pieces and equations to try and balance each of the formulas so that there is the same number of atoms on each side of the chemical reaction. Use the labels to label the reactants and products on your 2 equations.
The law of conservation of mass states that matter cannot be created or destroyed. It can only be rearranged to form a new substance. The two chemical equations each have an arrow in them. The arrow represents a chemical change. Use the pieces and equations to try and balance each of the formulas so that there is the same number of atoms on each side of the chemical reaction. Use the labels to label the reactants and products on your 2 equations.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Help
Transcribed Image Text:gs - Home
P Tavion Watson - Exploration - Bal x
E Tavion Watson - The Strangest, G X
+
/npatterson_mcpss_com/_layouts/15/Doc.aspx?sourcedoc=%7B66033F1F-0CC1-4FED-ADOC-4C8174139EBA%7D&file=Tavion%20Watson..
ing Chemical Equations Online - 2. 28- Saved
O Search
Single Line
sitions
Animations
Slide Show
Review
View
Help
Open in Desktop App
2 Share
O Comments
A A
A
Explore It! Part 1
KESLER
SCIENCE
The law of conservation of mass state
destroyed. It can only be rearranged to form a new substance.
that matt
cannot
created or
O2 2H2
The two chemical equations each have an arrow in them. The arrow represents
chemical change.
Use the pieces and equations to try and balance each of the formulas so that
there is the same number of atoms on each side of the chemical reaction.
Use the labels to label the reactants and products on your 2 equations.
Reactants
Reactants
→
Products
->
2H,0
Products
INPUT
STATION
Go to Part 2
Help Improve Office Notes 9
品豆
T
4:57 PM
1/6/2021
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

