The ligand substitution in organometallic complexes is very similar to that of coordination complexes. However, for organometallic complexes, the valence electron at central metal atom should not be more than 18. (a). Show TWO (2) steps synthesis of complex D by considering all of the following requirements: (i). CrO3 as a starting material and source of Cr (ii). only AsPh3 and CO as the ligand sources (can use more than 1 mole each) (iii). appropriate reaction conditions and solvents (iv). appropriate reagent(s) OC OC со Cr AsPh3 CO CO Complex D
The ligand substitution in organometallic complexes is very similar to that of coordination complexes. However, for organometallic complexes, the valence electron at central metal atom should not be more than 18. (a). Show TWO (2) steps synthesis of complex D by considering all of the following requirements: (i). CrO3 as a starting material and source of Cr (ii). only AsPh3 and CO as the ligand sources (can use more than 1 mole each) (iii). appropriate reaction conditions and solvents (iv). appropriate reagent(s) OC OC со Cr AsPh3 CO CO Complex D
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter14: Applications Of Ultraviolet-visible Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 14.7QAP: A 3.03-g petroleum specimen was decomposed by wet ashing and subsequently diluted to 500 mL in a...
Related questions
Question
1
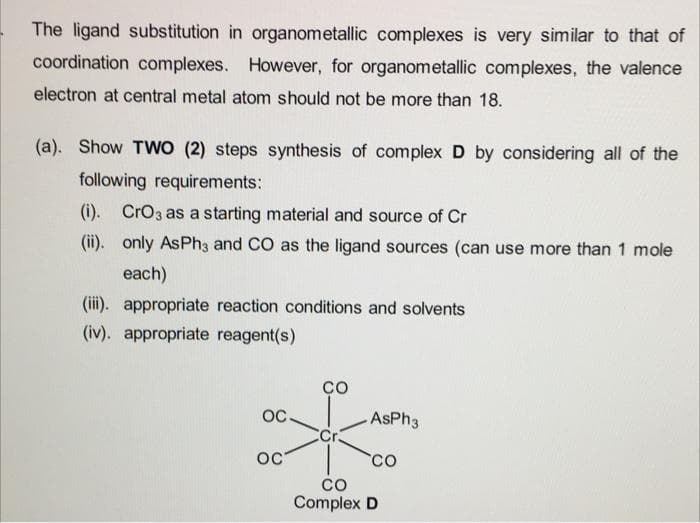
Transcribed Image Text:The ligand substitution in organometallic complexes is very similar to that of
coordination complexes. However, for organometallic complexes, the valence
electron at central metal atom should not be more than 18.
(a). Show TWO (2) steps synthesis of complex D by considering all of the
following requirements:
(i). CrO3 as a starting material and source of Cr
(ii). only AsPh3 and CO as the ligand sources (can use more than 1 mole
each)
(iii). appropriate reaction conditions and solvents
(iv). appropriate reagent(s)
OC
OC
со
Cr
AsPh3
CO
CO
Complex D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning