The mathematical expression for the change in free energy of a system is AG AH TAS %3D Which of the following statements is correct? Oa. When AG is a zero; AH = -T?S O b. AS is the change in enthalpy, a measure of randomness. O c. T is the temperature in degrees Celsius. O d. AH is the change in entropy, the energy available to do work.
The mathematical expression for the change in free energy of a system is AG AH TAS %3D Which of the following statements is correct? Oa. When AG is a zero; AH = -T?S O b. AS is the change in enthalpy, a measure of randomness. O c. T is the temperature in degrees Celsius. O d. AH is the change in entropy, the energy available to do work.
Chapter3: Ratio And Proportion
Section: Chapter Questions
Problem 1PP
Related questions
Question
None
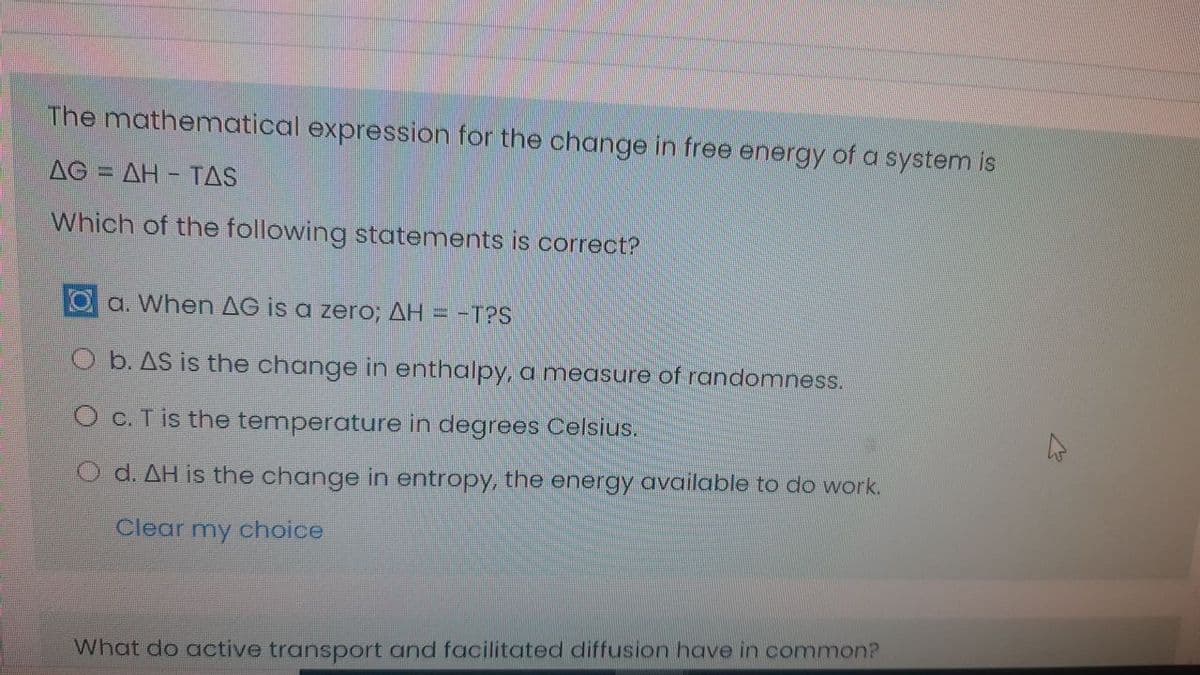
Transcribed Image Text:The mathematical expression for the change in free energy of a system is
AG = AH - TAS
%3D
Which of the following statements is correct?
a. When AG is a zero; AH = -T?S
O b. AS is the change in enthalpy, a measure of randomness.
O c. T is the temperature in degrees Celsius.
O d. AH is the change in entropy, the energy available to do work.
Clear my choice
What do active transport and facilitated diffusion have in common?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax


Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax